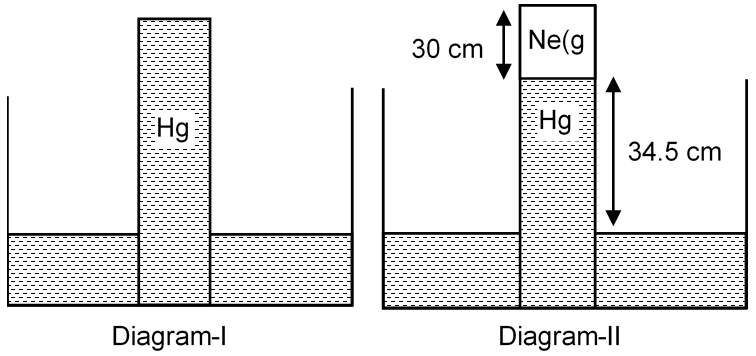
A barometer tube is put vertically down as shown in diagram (I). When a small amount of neon gas is introduced in the tube, it displaces the mercury column and occupy the space above the mercury column and the level decreases as shown in diagram (II). Calculate the mass (in mg) of the neon gas introduced.
Given: Room temperature = 27°C ; Atmosphere pressure = 1 atm
g = 10 m/s2 ; Cross section of tube = 2 cm2
density of Hg = 13.6 g/mL ; R = 8.3 J/mol. K
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
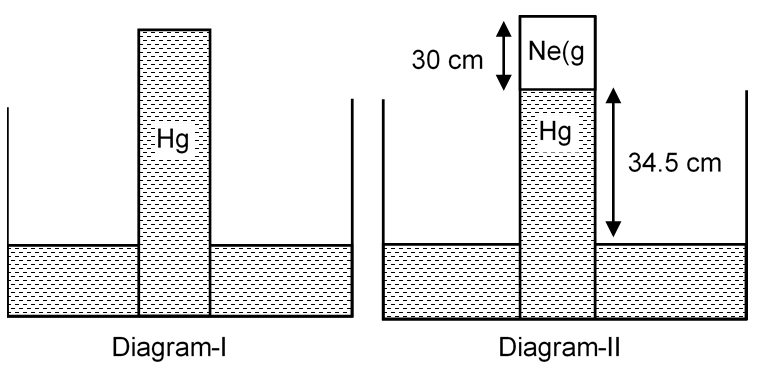
hNe column = 30 cm
Volume of Ne gas = 2 × 30 = 60 cm3 = 6 × 10–5 m3
Pressure of Ne gas = 76 – 34.5 = 41.5 cm of Hg
76 × dHg × g = 34.5 × dHg × g + PNe
PNe = (41.5 cm) (13.6 g/mL) (10 m/s2)
= (41.5 × 10–2 m) (13600 kg/m3) (10 m/s2) ⇒ 56440 Nm–2
n = = 1.36 × 10–3
wNe = 0.0272 g
= 2.72 × 10–5 kg