(a) Hydrocarbon 'A' gives monobromo derivative on reacting with excess HBr, 1 mole of the hydrocarbon require 6 moles of O2 for complete combustion, total number of carbon atoms present in hydrocarbon.
(b) Total number of possible isomers for 'A' are
(c) Hydrocarbon 'B' gives red ppt. with Cu2Cl2 NH4OH. One mole of 'B' require 8.5 moles of O2 for complete combustion. How many carbon atoms are present in the hydrocarbon.
(d) Position of multiple bond in hydrocarbon 'B' is
Write answer of part (a), (b), (c) & (d) in the same order and present the four digit number as answer. For example : If all these answer are 9 then fill 9999.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
(a)
3n = 6 × 2
C4H8 n = 4
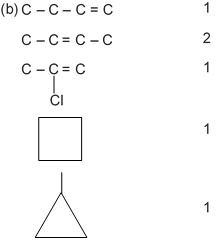
(c)
= 8.5 × 2
3n = 17 + 1
(d) C – C – C – C – C CH