Engineering
Chemistry
Quantum Numbers
Question
According to Aufbau principle, the correct order of energy of 3d, 4s and 4p-orbitals is:
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
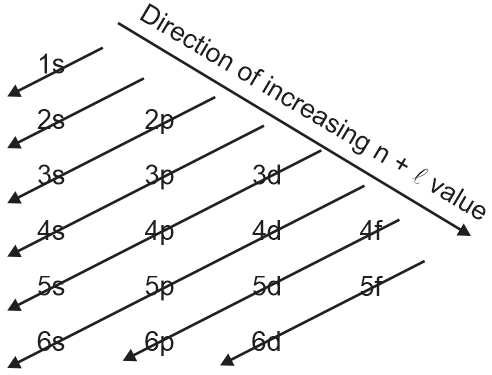
According to aufbau principle, the correct order of energy of 3d,4s and 4p-orbitals is \displaystyle 4s < 3d < 4p
Aufbau principle:
In the ground state of the atoms, the orbitals are filled with electrons in order of increasing energy.
The 4s sub-energy level is at a lower energy than the 3d sub-energy level
For 3d, n = 3 and ℓ = 2. Hence, (n + ℓ) = 3 + 2 = 5
For 4s, n = 4 and ℓ = 0. Hence, (n + ℓ) = 4 + 0 = 4
As the value of (n + ℓ) for 4s orbital is less than that of 3d orbital, 4s orbital is filled before 3d orbital.
For 4p, n = 4 and ℓ = 1. Hence, (n + ℓ) = 4 + 1 = 5
As the value of (n + ℓ) for 4p orbital is same as that of 3d orbital, 3d orbital is filled before 4p orbital as 3d orbital has lower value of n than 4p orbital.
Note: If two orbitals have the same value of (n + ℓ), then the orbital with the lower value of n will be filled first.
Aufbau principle:
In the ground state of the atoms, the orbitals are filled with electrons in order of increasing energy.
The 4s sub-energy level is at a lower energy than the 3d sub-energy level
For 3d, n = 3 and ℓ = 2. Hence, (n + ℓ) = 3 + 2 = 5
For 4s, n = 4 and ℓ = 0. Hence, (n + ℓ) = 4 + 0 = 4
As the value of (n + ℓ) for 4s orbital is less than that of 3d orbital, 4s orbital is filled before 3d orbital.
For 4p, n = 4 and ℓ = 1. Hence, (n + ℓ) = 4 + 1 = 5
As the value of (n + ℓ) for 4p orbital is same as that of 3d orbital, 3d orbital is filled before 4p orbital as 3d orbital has lower value of n than 4p orbital.
Note: If two orbitals have the same value of (n + ℓ), then the orbital with the lower value of n will be filled first.
Hence, the correct option is C