Engineering
Chemistry
Arrhenius Equation
Catalysis and Kinetics
Order of Reaction and Law of Mass Action
Question
Activation energy is:
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
Explanation:
The energy difference between the energy of reactant Er and threshold energy Et is activation energy as shown below figure:
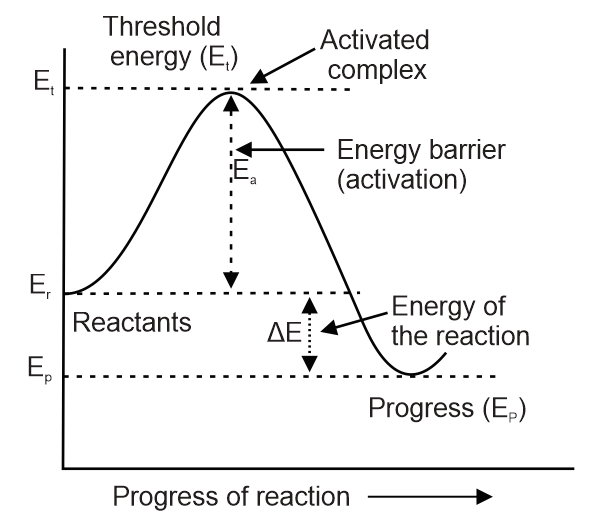
Therefore, option A is the correct answer.