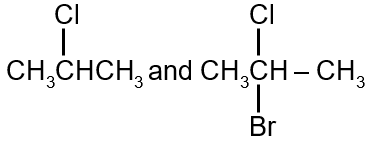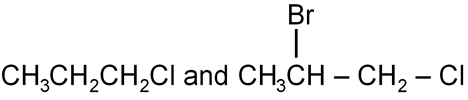Question
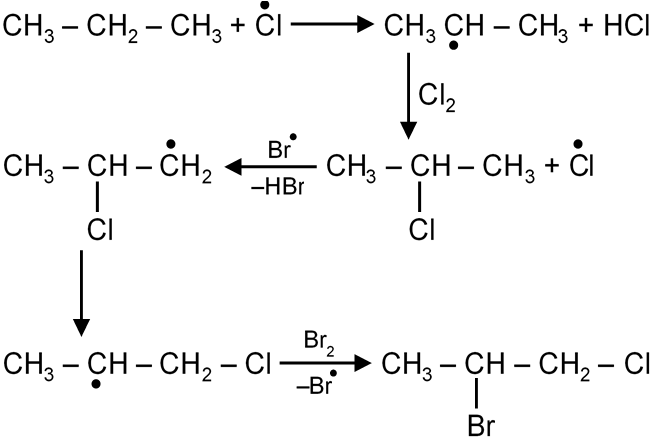
Alkanes are halogenated in presence of sunlight. The reaction follows free radical mechanism which completes in three steps.
(i) Chain initiation (fast)
(ii) Chain propogation (slow)
R°+X–X R–X + X°
(iii) Chain termination (fast)
R° + X° R – X
R°+ R° R – R
X° + X° X – X
Rate of this reaction increasing with stability of free radical formed and ease of its formation.
Rate 1° < 2°<3° (for same halogen)
Rate F2 > Cl2 > Br2 > I2 (for same alkane)
If excess of halogen is used one by one all H are replaced by X.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
A and B are:
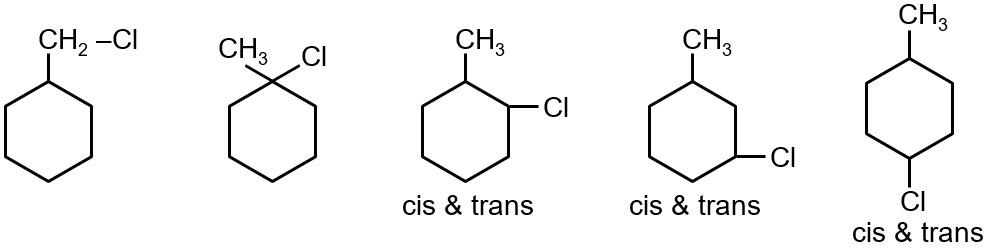
How many possible monochloro products (without counting mirror images) are formed when methyl cyclohexane is treated with Cl2 in presence of sunlight?
Correct order of rate of photochlorination for following compounds is:
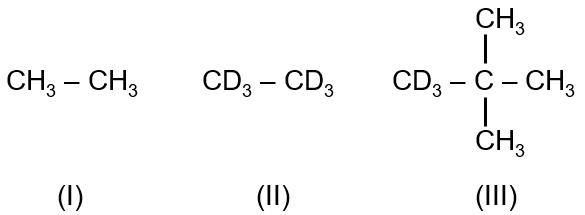
C–D bond is stronger than C–H bond. Neo pentane is 1.15 times more reactive than ethane towards photochlorination
Total number of chloro derivative possible for ethane is:
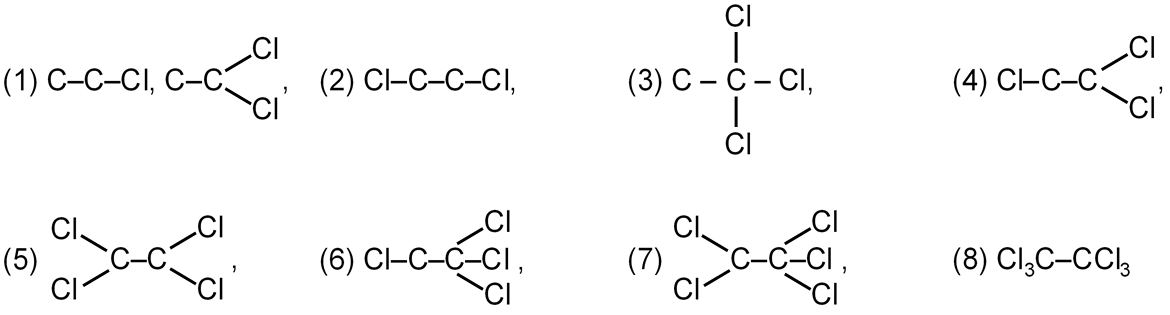
If rate determining step of this reaction is chain initiation not chain propogation what will be the order of rate of reaction for same alkane?
If chain initiation is RDS rate will be decided according to X–X bond energy order.