Question
An acid-base titration consists of the controlled adding of a dissolved base to a dissolved acid (or the reverse). Acid-base react rapidly to neutralize each other. At the equivalence point, enough titrant, the solution being added, has gone into make the chemical amounts of the acid and base exactly equal.
The pH of a titration changes every time a drop of titrant is added, but the rate of this change varies enormously. A titration curve, graph of pH as a function of the volume of titrant, displays in detail how the pH changes over the course of an acid-base titration. Significantly, the pH changes most rapidly near the equivalence point. The exact shape of a titration curve depends on the Ka and Kb acid and base.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
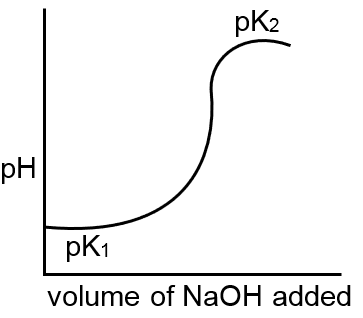
In the titration curve of HCl against NaOH, the pH equivalence point will be :
SA and SB titration equivalence point is always 7 because SASB salt have no hydrolysis
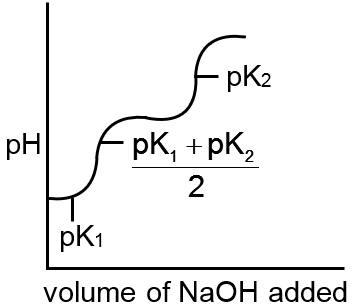
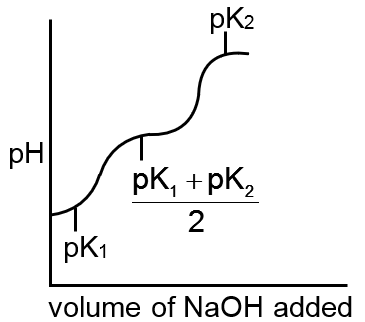
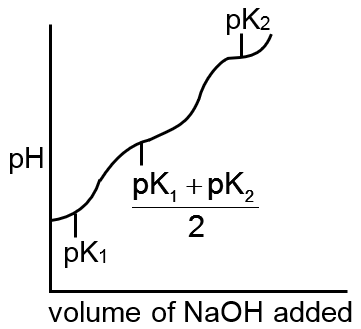
Which of the following curves indicates the titration of a weak diprotic acid by NaOH of equivalent
H2A + NaOH → NaHA + H2O first equivalence point
pH =
NaOH + NaHA → Na2A + H2O second equivalence point
pH = log C
sharp changes should be there at each equivalence point.

Which of the titrations also could it represent ?
If RbOH is titrated by HBr(SA) then curve goes down but if HBr is titrated by RbOH then curve goes up.