An experiment was conducted to determine the effect of concentration on the rate of diffusion. A student filled six dialysis bags with different concentrations of salt solutions and placed each of them in a separate beaker of distilled water.
Dialysis bags model the selective permeability of cell membranes and allow small molecules, ions and water to pass through according to the surrounding concentration gradient. A conductivity probe was used to measure the change in concentration of ions in the beaker over time.
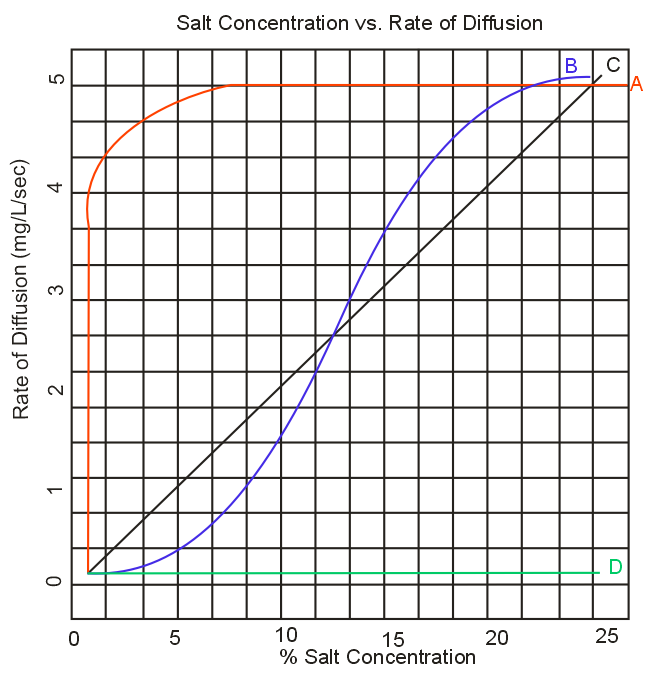
Which curve on the graph below accurately predicts the rate of diffusion as a function of salinity ?
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
The dialysis bag is placed in a hypotonic solution (distilled water), and not dialysing fluid for the passive movement of ions from the bag into the beaker. Even if the salt concentration is increased there would be movement of water from the beaker into the bag, but no movement of ions from the bag into the beaker. Therefore the rate of diffusion of ions will be near to zero