Engineering
Chemistry
Acid and Base GOC
Question
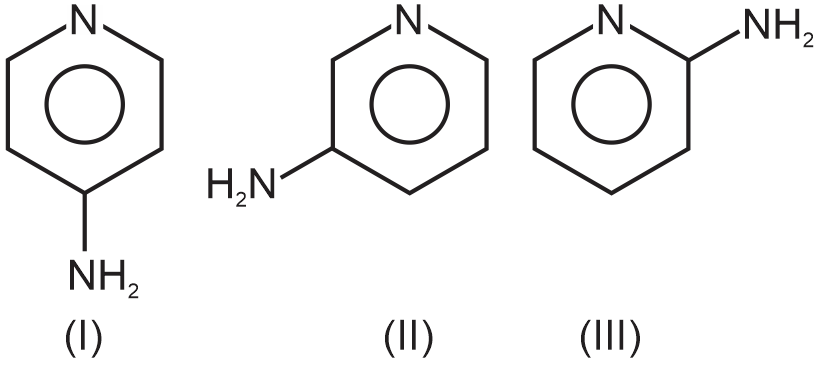
Basic strength order:
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
The decreasing order of basic strength is I > III > II.
Thus I is most basic and II is least basic. The lone pair on nitrogen atom of – NH2 group is in resonance with benzene ring. Due to this resonance, the electron density is maximum in ortho and para positions. Due to this, the basicity is maximum in I. The electron density is minimum in meta position. Hence, the basicity is minium in II. In the structure III, there is possibility of H bonding between ring nitrogen and the amino group. Hence, III is less basic than I.