Engineering
Chemistry
Structure of Some Important Compounds
Question
Blue vitriol has :
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
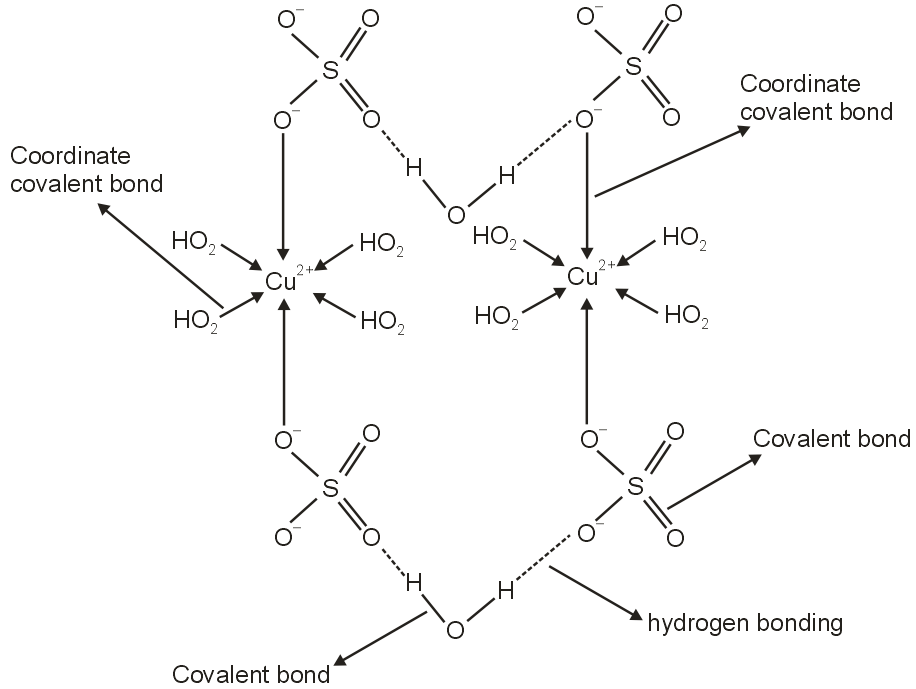
Blue vitriol or copper sulphate pentahydrate} has a molecular formula of CuSO4.5H2O of the five water molecules associated with this structure, only four are directly replaceable by NH3, which suggest that water molecule is somehow differently attached.
Blue vitriol is CuSO4.5H2O and it has all types of bonds.
The structure of CuSO4.5H2O in the solid-state is shown above
The Cu2+ ions are attracted towards SO42– ions not only by ionic interactions (electrovalent) but also by coordinate covalent bonds.
The Cu2+ ions form coordinate covalent bonds with water as well as sulfate ions.
There are covalent bonds in water and sulfate ions.