Configuration of
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
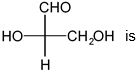
The configuration shown is a Fischer projection, a 2D representation of a 3D molecule. In this projection, horizontal lines represent bonds coming out of the plane (toward you), and vertical lines represent bonds going into the plane (away from you).
To assign the configuration, prioritize the four substituents by atomic number. The lowest priority group (often H) is on a vertical bond, meaning it is pointing away. The remaining three groups are traced from highest to second-highest to third-highest priority. If this sequence is clockwise, the configuration is R; if counterclockwise, it is S.
For this molecule, the order is -OH (1st), -COOH (2nd), -CH3 (3rd), -H (4th). The path 1→2→3 is counterclockwise. However, since the 4th priority is on a vertical bond (away), the direction is reversed. A counterclockwise path becomes R.
Final Answer: R