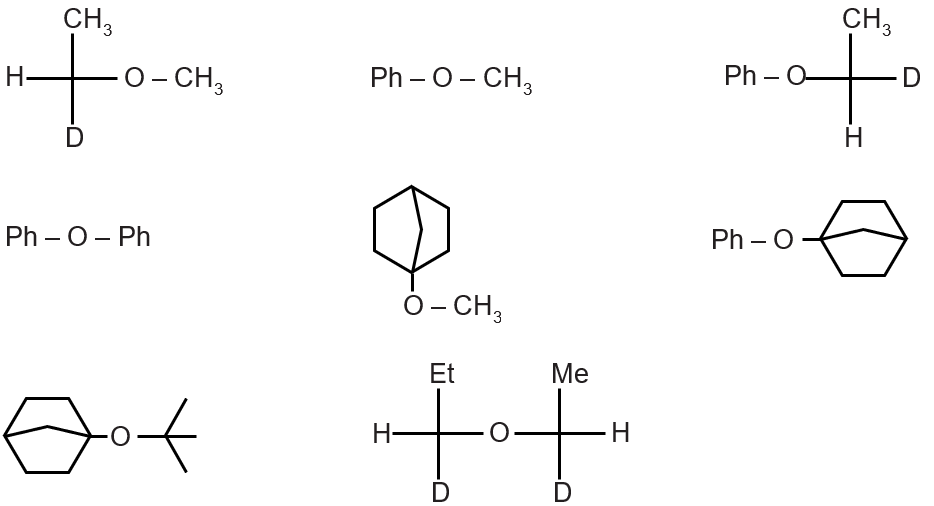
Consider following ethers and give number of ethers which can not be prepared by Williomson's ether synthesis.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Williamson's ether synthesis requires an alkyl halide and an alkoxide ion. It fails for tertiary alkyl halides due to elimination (E2) dominance over substitution. Also, it cannot form ethers where the alkoxide derived from a tertiary alcohol (bulky, prone to elimination).
Analyzing the structures:
Ether (a): Can be synthesized from CHC-ONa and CH (No β-hydrogens for elimination).
Ether (b): Cannot be synthesized. The required alkoxide is tertiary and bulky, and the alkyl halide (tertiary) will undergo elimination.
Ether (c): Can be synthesized from Ph-ONa and CH.
Ether (d): Cannot be synthesized. The alkyl halide component would be a tertiary halide, favoring elimination over substitution with any alkoxide.
Final Answer: 2