Engineering
Chemistry
Crystal Field Theory and Valence Bond Theory
Colour and Magnetism
Isomerism in Coordination Compounds
Question
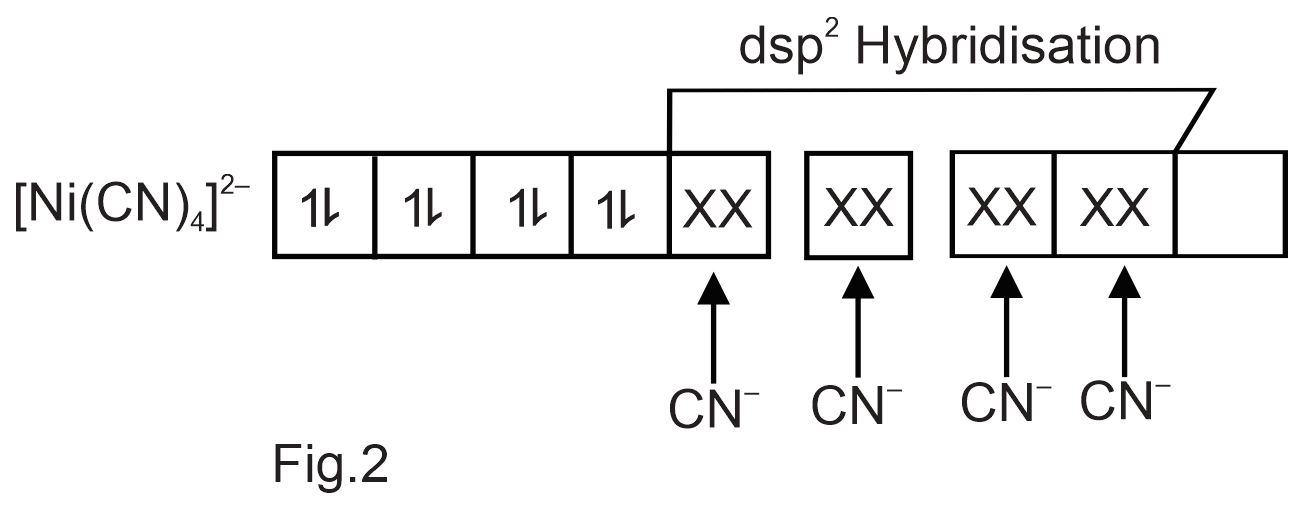
Explain the hybridization of the central metal atom in the complex [Ni(CN)4]2− with the help of diagram.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
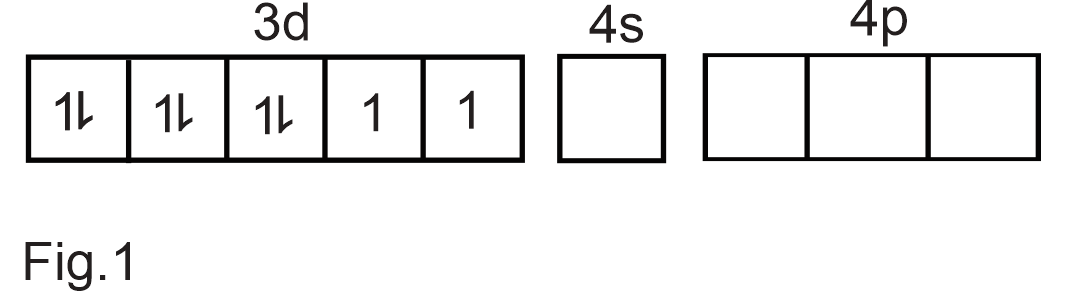
[Ni(CN)4]2− Electronic configuration of Ni:[Ar]4s23d8 Electronic configuration of Ni2+:[Ar]4s03d8 Outer electronic configuration of Ni2+ (Refer Fig.1) Pairing of electrons takes place in the presence of strong field ligand CN− Thus,one 3d orbital becomes vacant.(Refer Fig.2) As the hybridization involved is dsp2,we get square planar complex.As there are no unpaired electrons the complex is diamagnetic.