Engineering
Chemistry
Adsorption
Colloids Preparation and Properties
Hardness of Water
Question
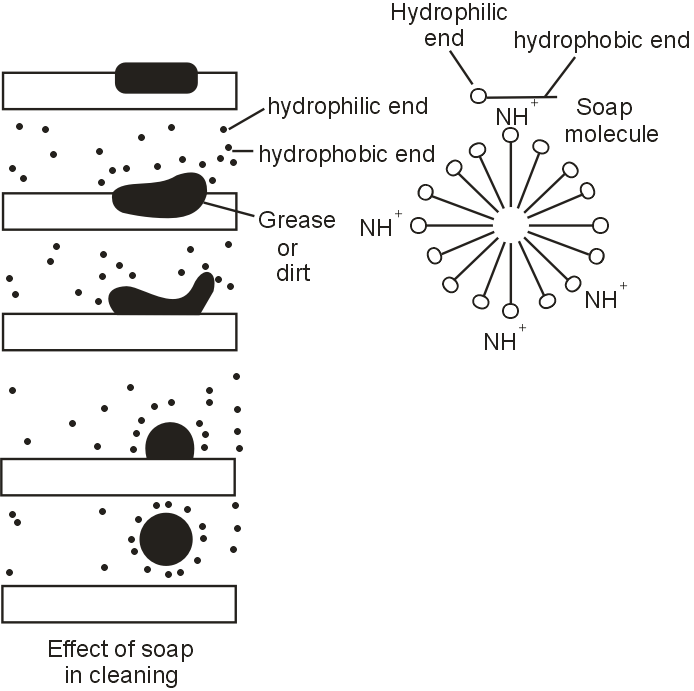
Explain the mechanism of the clearing action of soaps.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
Soap are molecules in which the two ends have differing properties. One is hydrophilic, that is, it interacts with water, while the other end is hydrophobic, that is, it interacts with hydrocarbons.
In the clusters of molecules in which the hydrophobic tails are on the surface of the cluster. This formation is called a micelle. Since the oily dirt will be collected in the center of the micelle.
The micelles stay in solution as a colloid and will not come together to precipitate because of ion-ion repulsion. Thus the dirt suspended in the micelles is also easily rinsed away.