Engineering
Chemistry
Polymers
Basic Concepts of GOC
Resonance Energy
Question
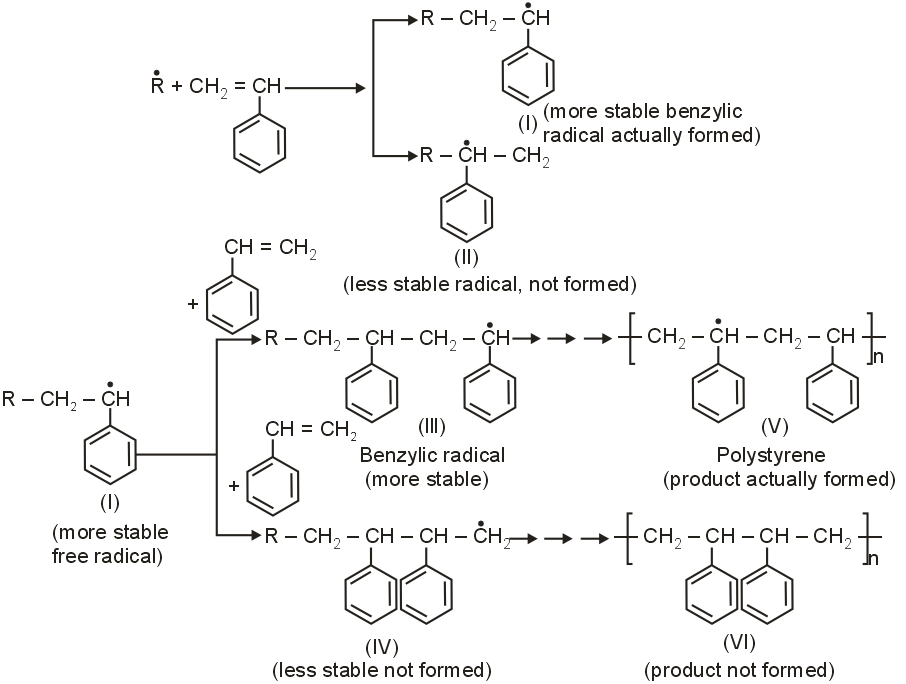
Explain why free radical polymerisation of styrene gives a product in which phenyl groups are on alternate carbon atoms rather than on adjacent carbon atoms.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
During free radical polymerisation, the addition of free radical to monomer molecules occurs in accordance with Markovnikov's rule so as to give more stable benzylic free radical.
For example,
This process goes on till polystrene (V) in which the phenyl groups are on alternate carbon atoms is obtained rather than the product (VI) in which the phenyl groups are on adjacent carbon atoms.