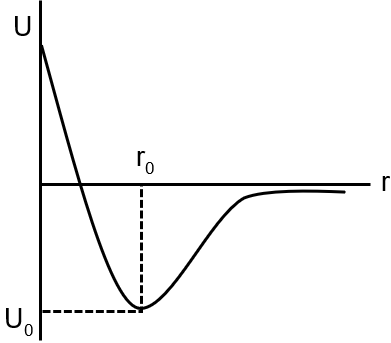
Figure is a graph of potential energy versus separation of molecules for a typical substance. Which of the following is a FALSE statement concerning this data ?
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
The potential energy (U) vs separation (r) graph shows the intermolecular interaction. At equilibrium separation r0, U is minimum and force F = -dU/dr is zero. The force is repulsive for r < r0 and attractive for r > r0. Kinetic energy is not maximum at r0; it depends on total energy. An ideal gas has no intermolecular forces, so its U=0 line would be flat, unlike this curve.
Evaluating the options:
The statement "at r0 the kinetic energy of molecule is maximum" is FALSE. At r0, potential energy is minimum, but kinetic energy depends on the total energy of the system and is not necessarily at a maximum. For a bound system, kinetic energy is actually often higher when potential energy is higher (e.g., in a simple harmonic oscillator, KE is max at equilibrium).
Final Answer: at r0 the kinetic energy of molecule is maximum.