Find out number of reagents which can oxidise 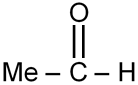
(i) I2/NaOH
(ii) KMnO4/H⊕
(iii) Tollen's reagent
(iv) LiAlH4
(v) NaBH4
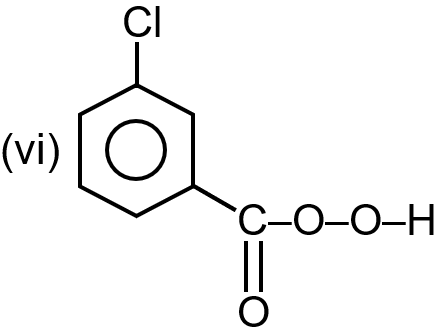
(vii) SeO2/Δ
(viii) NH2 – NH2 / pH (4–6)
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
The compound is an aldehyde. Oxidizing agents convert aldehydes to carboxylic acids. Reagents (i) I₂/NaOH (Haloform reaction oxidizes methyl ketones, not aldehydes), (ii) KMnO₄/H⁺ (oxidizes aldehydes), (iii) Tollen's reagent (oxidizes aldehydes), (iv) LiAlH₄ (reducing agent), (v) NaBH₄ (reducing agent), (vi) SeO₂/Δ (oxidizes methylene groups, not aldehydes), (vii) NH₂–NH₂/pH 4-6 (reducing agent). Only (ii) and (iii) are oxidizing agents that work on aldehydes.
Final Answer: 2