Engineering
Chemistry
Ionisation Electron Gain Atomisation enthalpy
Question
Find the Bond enthalpy (in kJ/mol) of one "three centre two electron bond" in B2H6
{B–H–B → 2B(g) + H(g)} from the given data.
ΔHf° [BH3(g)] = 100 kJ/mol ΔHf° [B2H6(g)] = 36 kJ/mol ΔHf°
ΔHatm [B(s)] = 565 kJ/mol ΔHatm = [H2(g)] = 218 kJ/mol
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
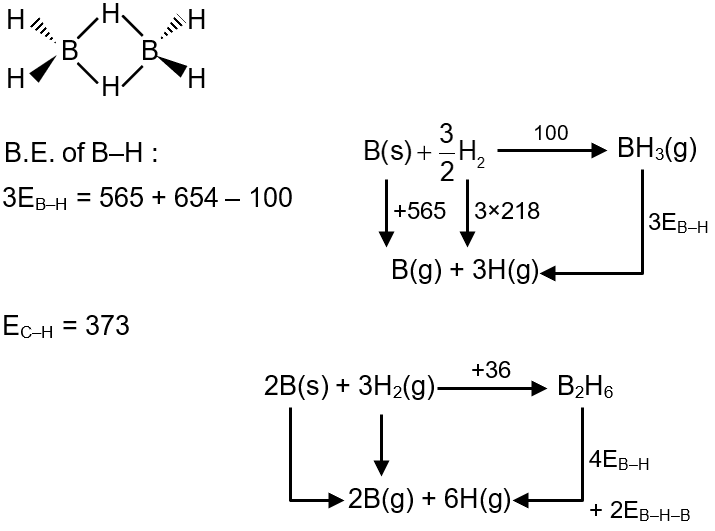
2EB–H–B = 2 × 565 + 6 × 218 – 36 – 4 × 373
EB–H–B = 455 kJ/mol = 4.55 × 102 kJ/mol