For an endothermic reversible reaction
4A(g) + B2(g) ⇌ 2A2B(g)
Neglect the slope of change in concentration when system approaches equilibrium.
Assume that changes are carried out very fast.
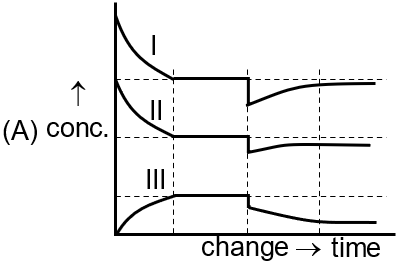
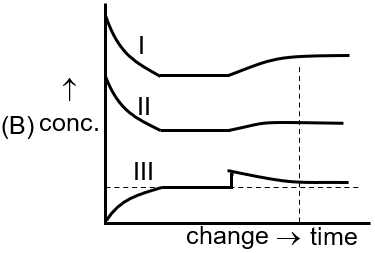
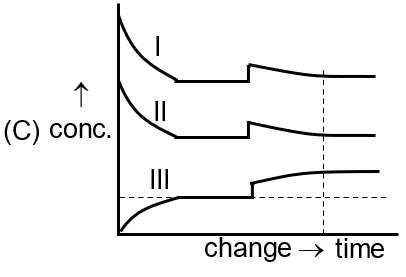
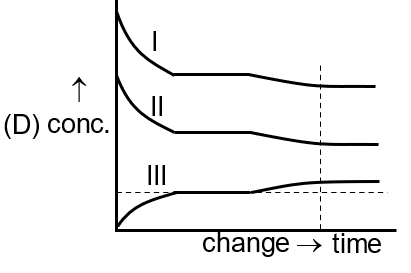
[Curve I for A(g) , Curve II for B2 (g), Curve III for A2B(g)]
| Column-I | Column-II |
 |
(P) Increase in temperature |
 |
(Q) Increase in pressure |
 |
(R) Addition of A2B at equilibrium |
 |
(S) Addition of inert gas at constant pressure |
| (T) Increase of volume. |
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
This question involves matching disturbances (Column-II) to concentration-time graphs (Column-I) for the endothermic reversible reaction: 4A(g) + B₂(g) ⇌ 2A₂B(g). The key is to apply Le Chatelier's Principle to predict how the equilibrium shifts and how concentrations change instantly and at the new equilibrium.
For an endothermic reaction, increasing temperature (P) favors the forward reaction. The graph shows a sharp decrease in reactants (Curves I & II) and a sharp increase in product (Curve III), followed by a new equilibrium, matching the first image.
Increasing pressure (Q) or decreasing volume (T) shifts equilibrium toward the side with fewer moles of gas. Here, the product side has fewer moles (2 vs 5), so [A₂B] increases. The second image shows this for Curve III.
Adding product A₂B (R) shifts equilibrium left, increasing reactants and decreasing product. The third image shows a sharp rise in [A₂B] followed by a decrease, and a sharp drop in reactants followed by an increase.
Adding inert gas at constant pressure (S) increases total volume but not partial pressures, so no shift occurs; concentrations remain unchanged, as shown by the flat lines in the fourth image.
Final Answer: 1-P, 2-Q/T, 3-R, 4-S