Engineering
Chemistry
Raoult Law and Vapour Pressure
Question
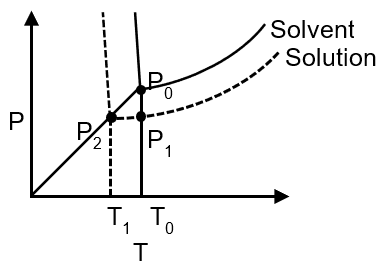
From the phase diagram of water and an aqueous solution containing non volatile solute, identify the incorrect option:
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
(A) Temperature T0 , represent tripple point of solvent where
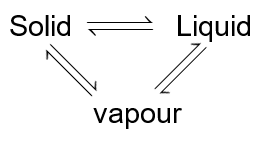
are in equilibrium hence vapour pressure of solid and vapour pressure of liquid will be same.
(B) Figure showing P0 > P1 > P2
(C) Line corresponding to P0 and P2 vapour pressure
Hence ln
(D) Line corresponding to P1 and P2 vapour pressure
Hence ln