Engineering
Chemistry
Hydrogen Bonding
Boiling point
Basic Concepts of GOC
Question
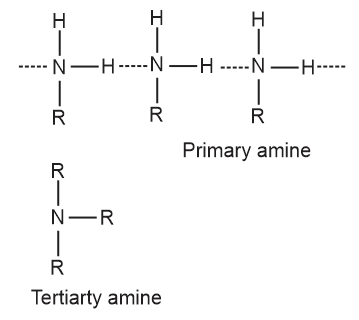
Give plausible explanation for: Why do primary amines have higher boiling points than tertiary amines?
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
Primary amines are engaged in intermolecular association due to hydrogen bonding between nitrogen of one and hydrogen of another molecule.
Tertiary amines do not have intermolecular associations due to the absence of hydrogen atom available for hydrogen bond formation.
Therefore, primary amines have higher boiling points than tertiary amines.