Foundation
Chemistry Foundation
Allotropes of Carbon
Question
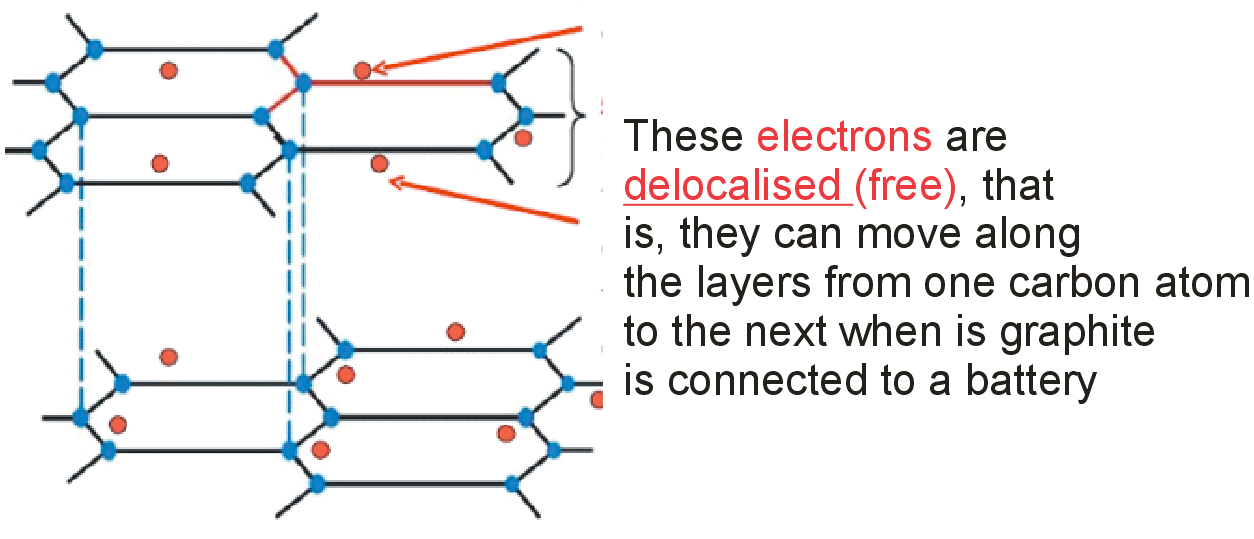
Graphite is a conductor of electricity because
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
Graphite is a conductor of electricity because as can be seen in the crystal structure of graphite (in the figure) it has free valence electrons which can conduct electricity.
Graphite displays the property of both metal and non-metal. As a metal, graphite is a good conductor of electricity due to the mobility of electrons in outer valence shells.