Engineering
Chemistry
Classification of Chemical Reaction
Oxidation Number and Calculation
Hydrogen Peroxide Properties and Preparation
Question
(i) Draw the gas phase and solid phase structure of H2O2.
(ii) H2O2 is a better oxidising agent than water. Explain.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
(i)
(ii) In H2O2 oxygen is in -1 oxidation state which has tendency to become -2 that is why it is better oxidising agent than water.
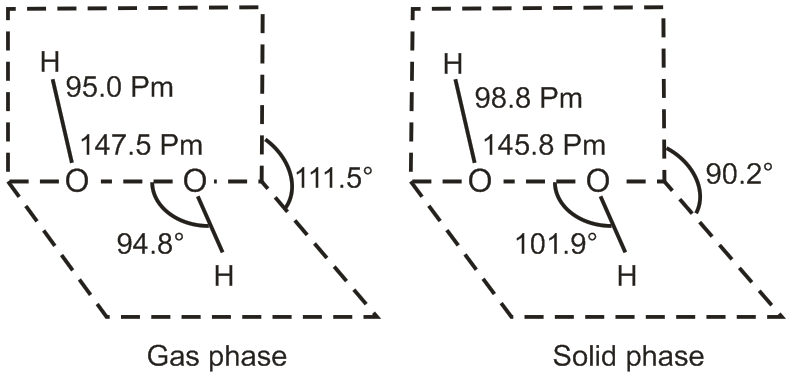
(a) H2O2 structure in gas phase, dihedral angle is 111.5°
(b) H2O2 in solid phase at 110 K, dihedral angle is 90.2°