Engineering
Chemistry
Enthalpy of Neutralisation and Resonance Energy
Question
If enthalpy of hydrogenation of C6H6(ℓ) into C6H12(ℓ) is –205 kJ & resonance energy of C6H6(ℓ) –152 kJ/mol then enthalpy of hydrogenation of 1,4-cyclohexadiene (ℓ) is
[Assume ΔHvap of C6H6(ℓ), C6H8(ℓ), C6H12(ℓ) all are equal]
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
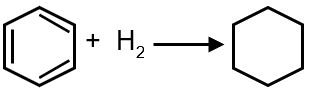
= ΔHT – ΔHA = – 152
= ΔHT = – 152 – 205 = – 357
![]()
![]()
ΔH = + 2 × (–119) = – 238 kJ/mol