Engineering
Chemistry
Ellingham Diagram
Question
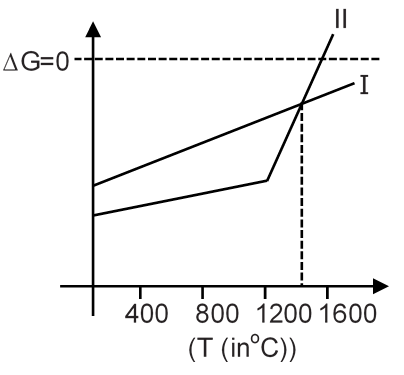
If ΔG vs T graph of
I → Al + O2 → Al2O3
II → 2Mg + O2 → 2MgO
is given as below :
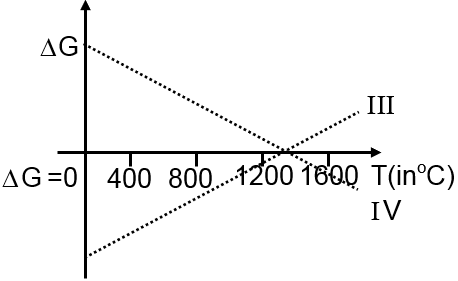
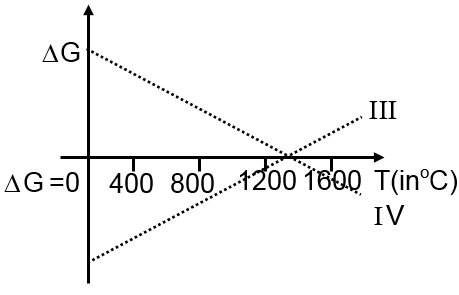
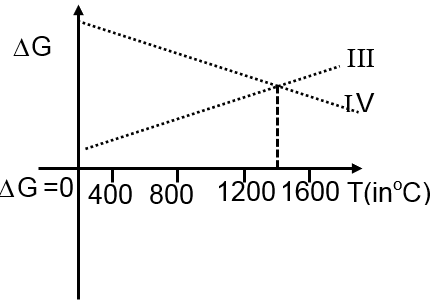
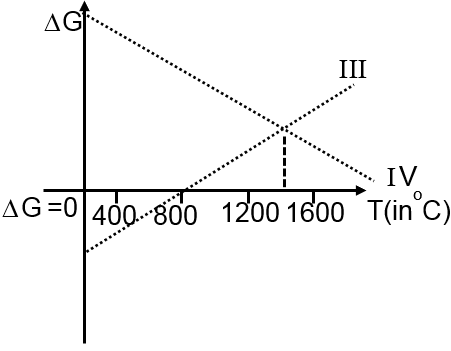
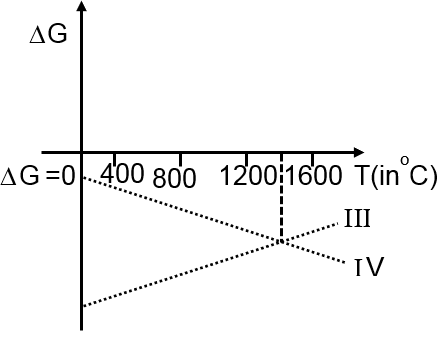
Then ΔG vs T graph of reaction
III 3Mg + Al2O3 3MgO + 2Al
IV 3MgO + 2Al 3Mg + Al2O3
are:
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
at T < 1400°C at T > 1400°C
III 3Mg + Al2O3 3MgO + 2Al G < 0 G > 0
IV 3MgO + 2Al 3Mg + Al2O3 G > 0 G < 0