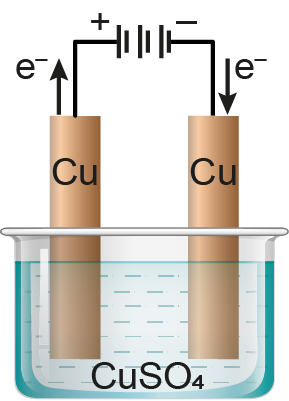
In the adjacent diagram the electrolytic cell contains 1 L of an aqueous 1 M Copper (II) sulphate solution. If 0.4 mole of electrons are passed through the cell, the molar concentration of copper ion after passing of the charge will be :
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
An electrochemical cell is a cell that can be created by placing mettalic ellectrod into an electrolyte where chemical reaction either uses or generates qusent
For eq = galvanic cell
having the redox reaction a galvanic cell arises the enengy transfer between electrons to convert chemical energy into electrical energy A galvanic cell can be made out of any two metals can form the anode and cathode if left in contact with each others.