Engineering
Chemistry
Acid and Base GOC
Question
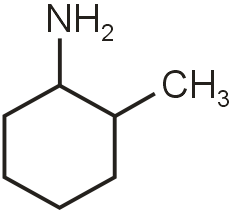
In the given pair identify most acidic compound in (A) and (B). Most basic in (C) and (D).

Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
Acidity increases with electron withdrawing group. Ortho effect increases acidity. Basicity increases with electron donating group. Ortho effect decreases basicity.
So. in case of
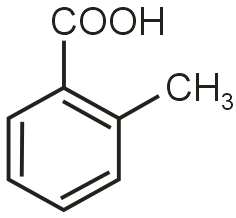
(Ortho effect)
in case of B most acidic is
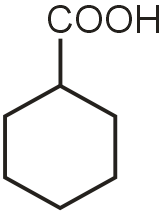
in case of C most basic is
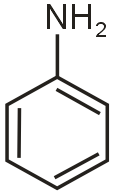
in case of D most basic is