Engineering
Chemistry
Vander Waal Gas and compressibility Factor
Liquefaction and Critical Temperature Pressure and Volume
Pressure Volume and Temperature
Question
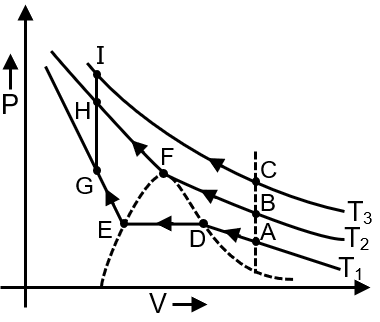
Isotherms of a real gas is represented as shown in diagram at three temperature T1, T2, T3. From the graph, predict in which case gas-liquid transition will involve only one phase throughout.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
ADEG and BADE involve two phase equilibrium along DE
BCI involves no phase transition,
In, ACIH ⇒ at A, C, I only gaseous phase
Just above point H ⇒ Gaseous phase
Just below point H ⇒ Liquid phase