Engineering
Chemistry
Alkane
Question
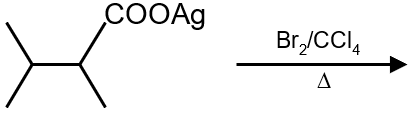
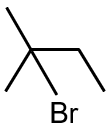
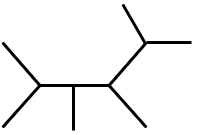
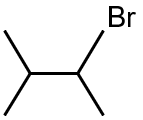
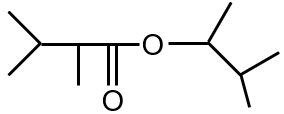
Major product is :
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
The reaction involves the addition of HBr to an alkene, following Markovnikov's rule. The hydrogen adds to the less substituted carbon, and the bromine adds to the more substituted carbon. The given alkene is symmetric, so both carbons of the double bond are equivalent. Therefore, the major product is 2-bromopropane.
The chemical equation is:
Final Answer: The first option (2-bromopropane) is the major product.