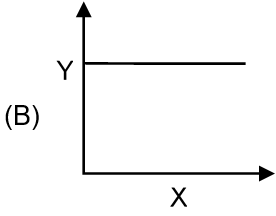
Match the graphs given in column I to the parameters and conditions.
{Given : ROR represents rate of reaction , [R] represents concnetration of reactant, t1/2 represents half life, [R]0 represents initial concentration of reactant }.
All other units have their usual meaning. Assume reactions to involve only one reactant unless otherwise stated.
| Column I | Column II |
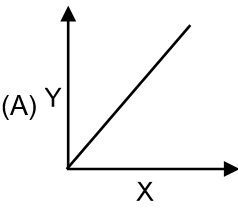 |
(P) Graph of lnKeq vs for exothermic reactions with = 0 and no entropy (ΔnCPm)Rxn change |
 |
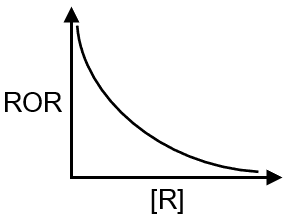
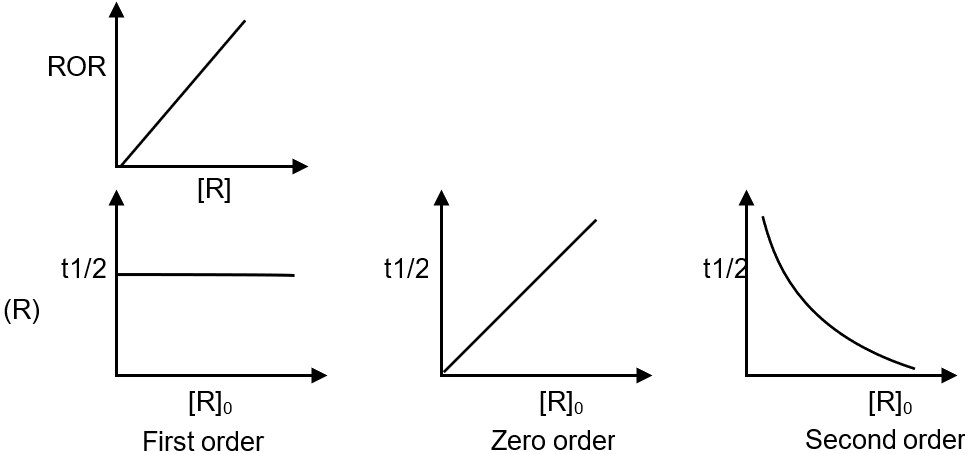
(Q) Graph of ROR vs [R] for a first order reaction |
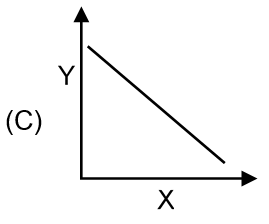 |
(R) Graph of t1/2 vs [R]0 for ether zero order, first order or second order. |
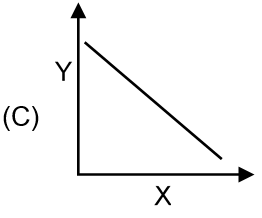 |
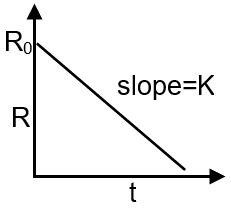
(S) Graph of [R] vs time for a zero order reaction. |
| (T) Graph of rate of reaction vs [R] for a (–1) order reaction. |
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
(P) lnKP vs Exothermic reaction : ΔH° = – ve ,ΔS° = 0
ΔG° = – RT ln KP
ΔH° – TΔS° = – RT ln KP
ΔH° = – RT ln KP
ln KP = –
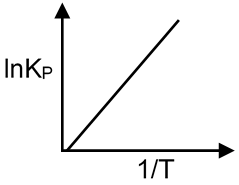
As ΔH° = – ve slope = +ve
(Q) ROR = K [R]' first order reaction
(S) R0 → R = Kt zero order reaction
R = R0 – Kt
(T) Rate of reaction = K[R]–1
ROR × [R] = K