Engineering
Chemistry
Dichromate and Permanganate Ion Reactions
Isomerism in Coordination Compounds
Resonance
Question
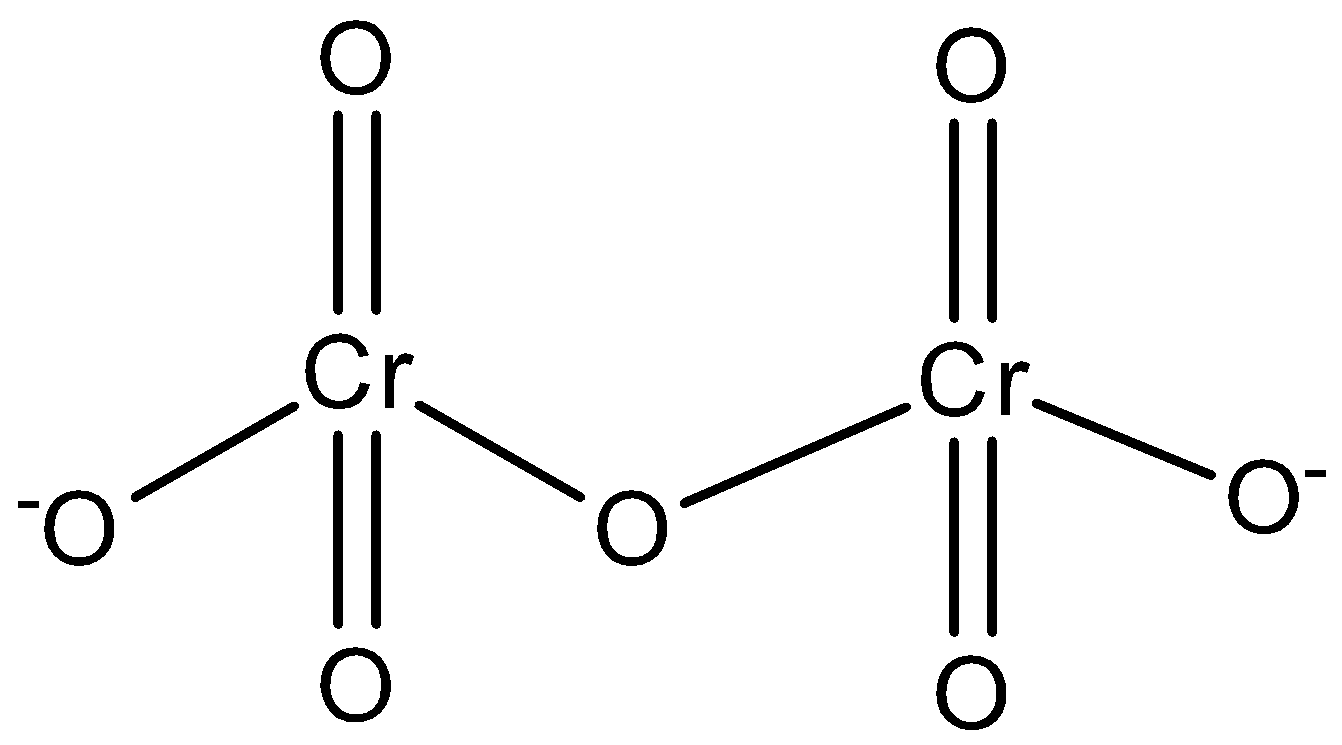
Number of identical Cr-O bonds in dichromate ion Cr2O72− is :
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
The correct answer is - There are '6' identical Cr – O bonds in the dichromate ion (Cr2O7–2)
In a dichromate ion (Cr2O7–2), there are 6 terminal Cr – O bonds. All these bonds are equivalent due to resonance.
The Dichromate ions (Cr2O7–2) are oxoanions of chromium in the oxidation state +6.
Hence option B is correct answer.