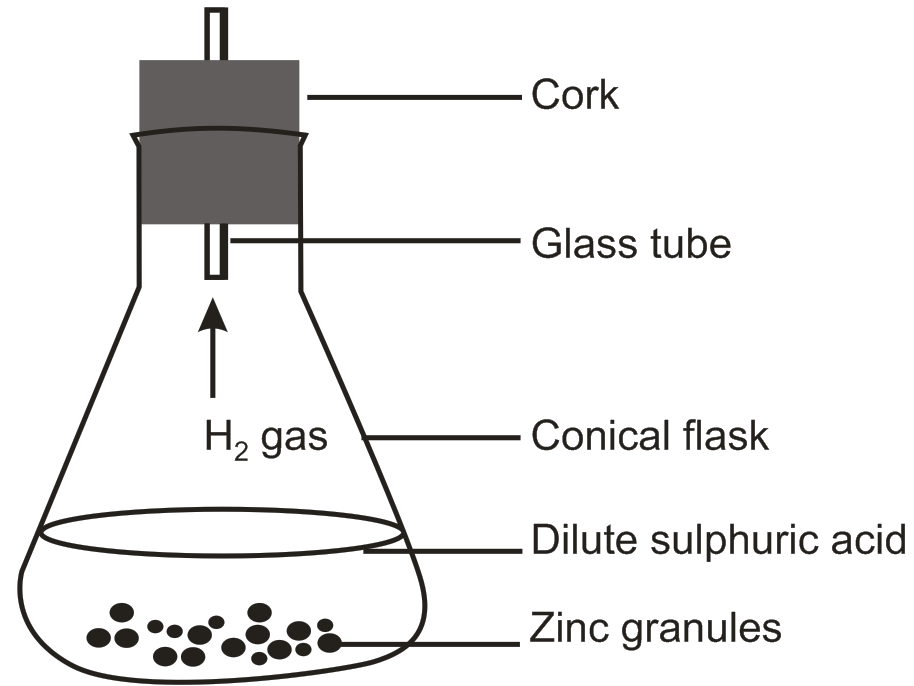
(a) Do you observe anything happening around the zinc granules?
(b) Is there any change in its temperature?
(c) Why is glass tube not dipped in dil H2SO4?
(d) How is H2 gas collected by downward displacement or upward displacement of water?
(e) Is H2 gas soluble or insoluble in water?
(f) Is H2 gas heavier or lighter than air?
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Zn + H2SO4 → ZnSO4 + H2 ↑
(*) We can observe bubbles of H2 gas being formed around zinc granules.
(*) This is an exothermic reaction, i.e. it involves release heat and the flask become hot and temperature of the system increases.
(*) The glass tube is not dipped in the solution so as to collect enough, H2 gas. It is was dipped in H2SO4 solution would enter the tube presenting H2 gas to enter the tube.
(*) H2 gas is collected by downward displacement of water.
(*) H2 gas is insoluble in water.
(*) H2 gas is lighter than air.