Engineering
Chemistry
Solubility Chart
Question
Salt (X) gives white ppt. with BaCl2 which is insoluble in conc. HNO3 and conc.HCl. Salt (X) on reaction with NaOH in presence of H2O2 gives green solution (Y) which on acidification turns into purple coloured compound (Z).
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Linked Question 1
The ratio of p bonds in compound (Y) and (Z) respectively is
Solution
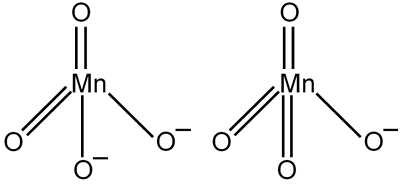
Linked Question 2
Compound (Y) is
Solution
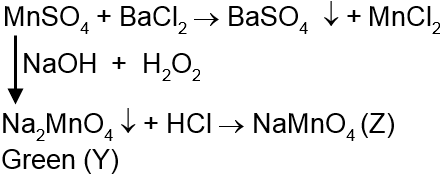
Y =
= d1 (paramagnetic with 1 unpaired electron)
Linked Question 3
Anions of compounds X, Y and Z are :
Solution
No Solution