Engineering
Chemistry
Conformational Isomerism
Alkene
Electrophilic addition
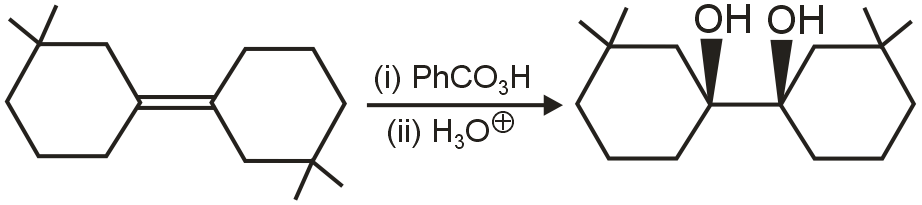
Question
Statement - 1 : The product of the reaction is optically inactive.
Statement - 2 : In reactions involving epoxidation followed by hydrolysis, the overall addition is anti-oriented.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
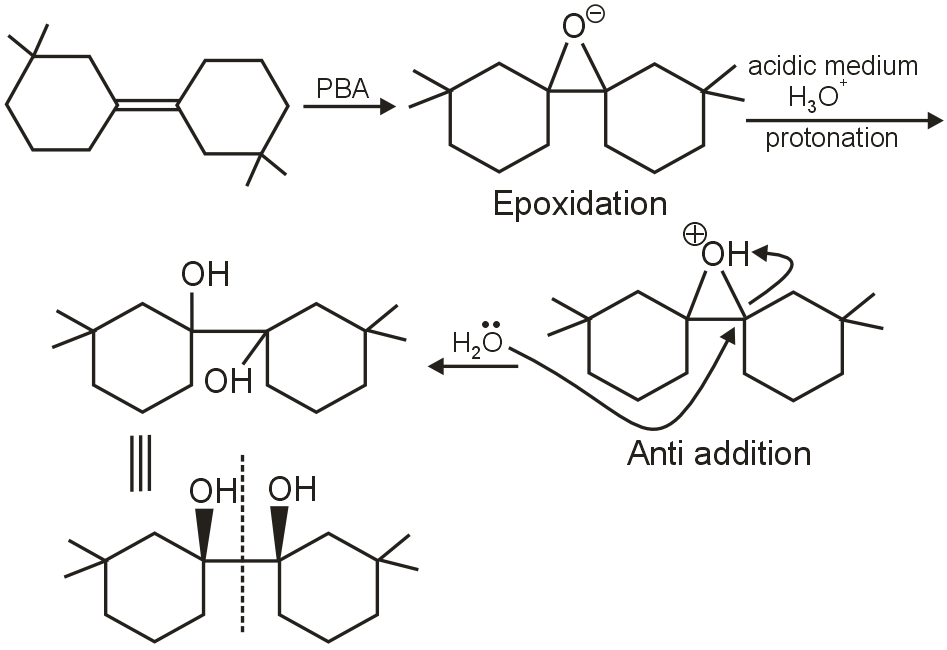
optically inactive - due to plane of symmetry.
Addition is anti - oriented.
So. Statement I : Product of reaction is optically inactive
Statement II : In reaction involving epoxidatios followed by hydrolysis, the onciall addition is Anti oriented.