Engineering
Chemistry
Calculation of Various Parameters at Equilibrium
Order of Reaction and Law of Mass Action
Arrhenius Equation
Question
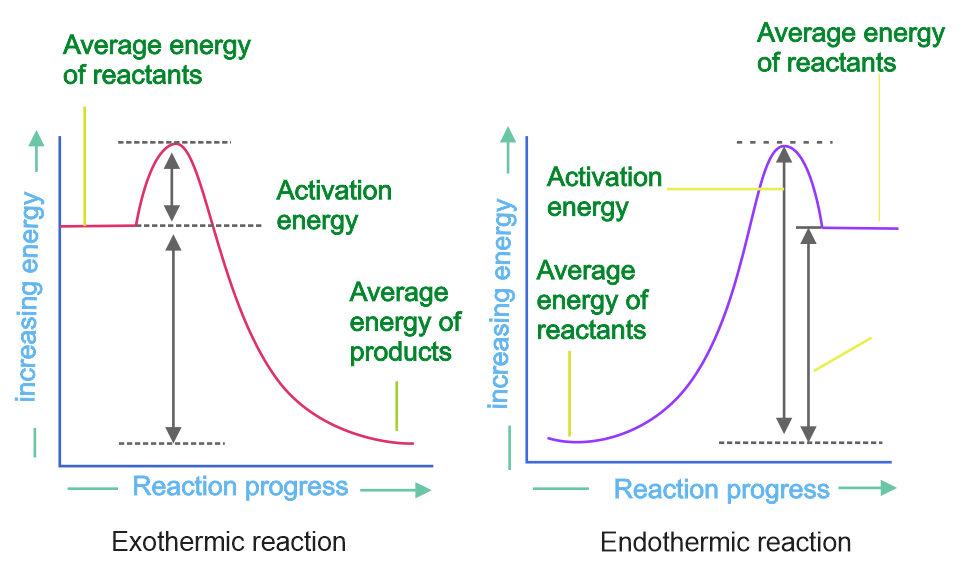
The activation energy for a simple chemical reaction A → B is Ea in the forward reaction. The activation energy of the reverse reaction:
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
As we know,
For exothermic reaction,
ΔHº = (PE of product) - (PE of reactant) < 0.
(Ea)f, < (Ea)b.
For endothermic reaction:
(Ea) < (Ea)b.