Engineering
Physics
Degree of Freedom Internal Energy of Gas and Specific Heat Capacity of Gases
Thermodynamics
Cyclic Processes
Question
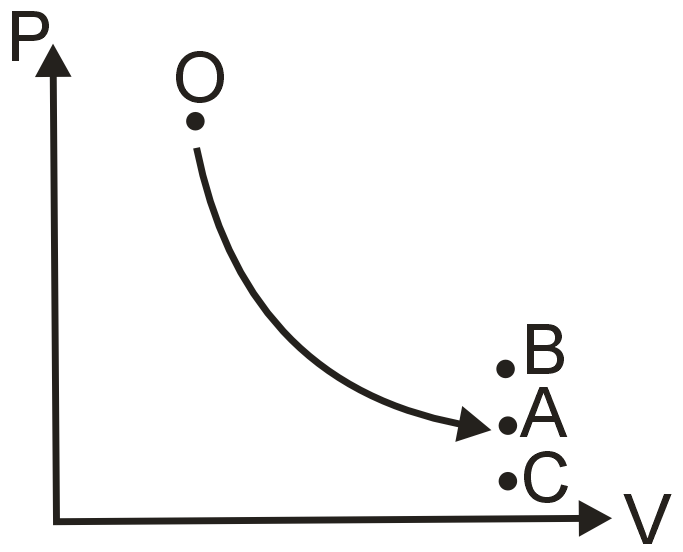
The arrow OA in the PV diagram shown in figure represents a reversible adiabatic expansion of an ideal gas. The same sample of gas, starting from the same state O, now undergoes a process to the same final volume. Select the appropriate combination of process and final state.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution