Question
The complex (A) is formed when FeSO4 reacts with excess of KCN. Complex (A) is oxidised into complex (B) by suitable oxidizing agent. From complex (B) three ligands are replaced by another three ligands (all are of same type and nonambidentate ligand) to form complex (C). From complex (B) when 4 ligands are replaced by another 4 ligands (of same type and nonambidentate ligand) it forms complex (D).
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
E.A.N. of complex (A) is :
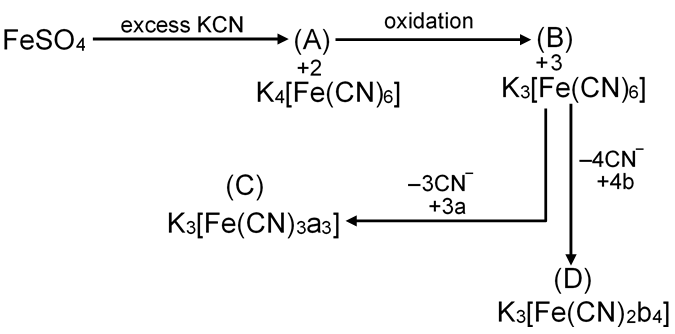
EAN = 26 – 2 + 6 × 2 = 36
How many total possible isomers we get from complex (D)?
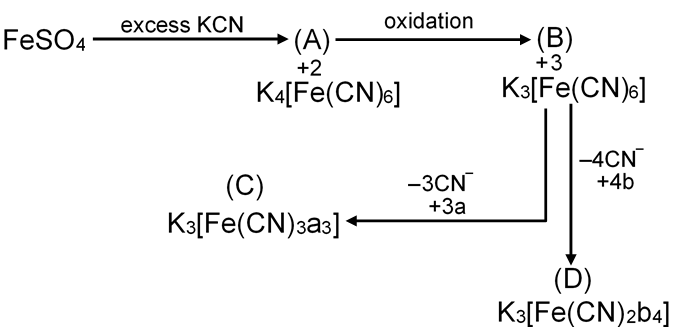
K3[Fe(CN)2b4]
| CN– | NC– | b | G.I. | |
| 2 | 0 | 4 | [Ma4b2] | 2 |
| 1 | 1 | 4 | [Ma4bc] | 2 |
| 0 | 2 | 4 | [Ma4c2] | 2 |
| _______________________________________ Total isomers = 6 _______________________________________ |
||||
How many total possible geometrical and structural isomers we get from complex (B)?
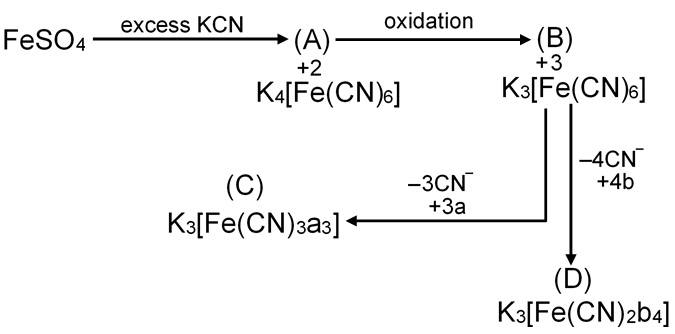
K3 [Fe(CN)6]
Total possible geometrical and structural isomers = 10