Engineering
Chemistry
Development of Periodic Table
Question
The diagram below shows part of the skeleton of the Periodic Table in which elements are indicated by letters which are not their usual symbols
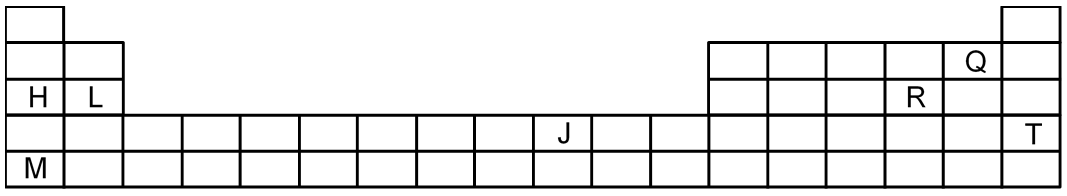
Which one of the following statements is correct?
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
M = Alkali metal (Rb) , Q = Halogen (F)
Therefore, M2Q will not exist
J is a transition element precisely it will be Ni hence Ni++ is coloured due to presence of unpaired electron.
All alkali carbonates are water soluble.
R cannot be gas at room temperature, it will be Sulphur element