Engineering
Chemistry
Preparation of Amines
Chemical Reactions of Amines
Ammonia
Question
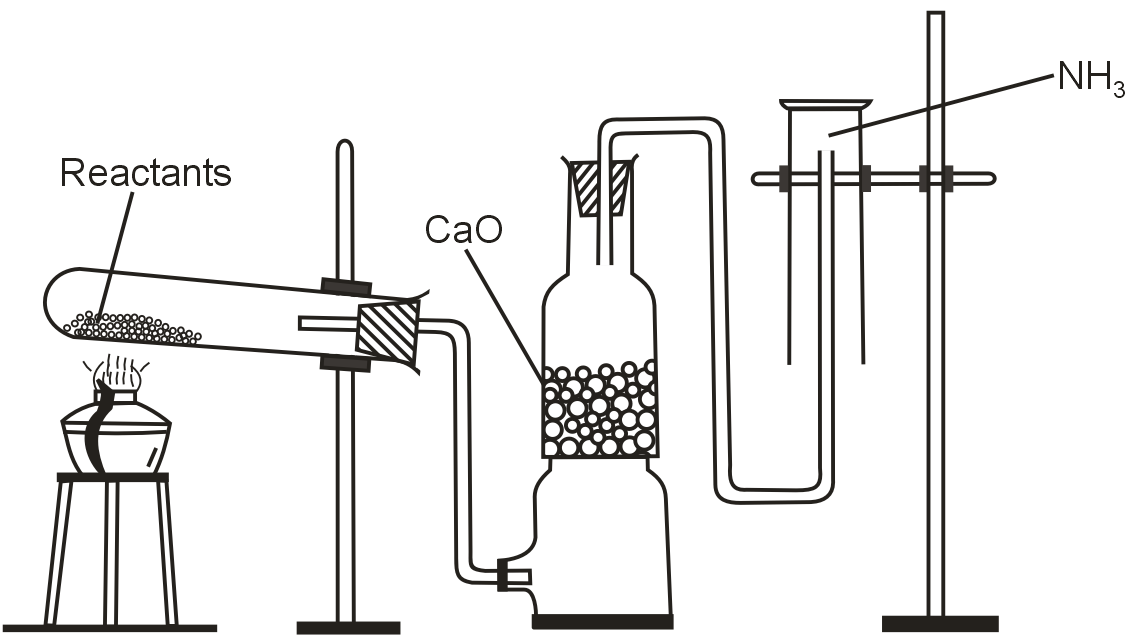
The diagram showing the laboratory preparation of NH3 is given.
(a) What are the reactants used ?
(b) Why CaO is used ?
(c) What is the reason behind not keeping the jar in the upward direction ?
(d) Write an experiment to identify presence of NH3 in the gas jar.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
(a) Ammonium chloride and calcium hydroxide
(b) To get dry ammonia, the gas is passed through drying the tower containing lumps of CaO.
(c) It is collected by the downward displacement of air because it is lighter than air.
(d) Fountain experiment
Being basic, ammonia gas changes red litmus solution blue.