Engineering
Chemistry
Crystal Field Theory and Valence Bond Theory
Colour and Magnetism
Isomerism in Coordination Compounds
Question
The geometry of [Ni(CN)4]2– and [NiCl4]2– are :
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
⇒ [Ni(CN)4]2–
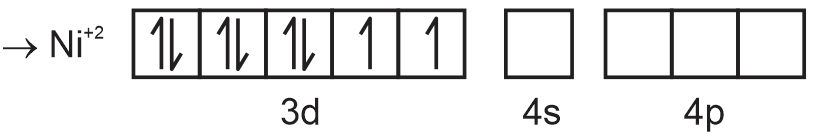
→ When CN– ligand approaches pairing takes place.
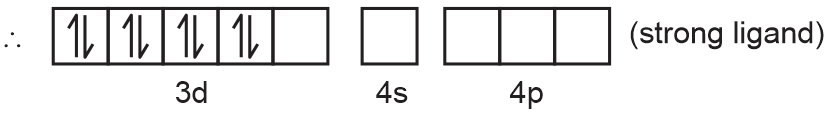
→ Ni+2 in [Ni(CN)4]–2
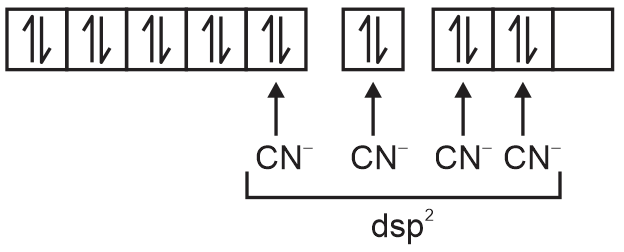
Geometry = square planer.
⇒ [NiCl4]–2
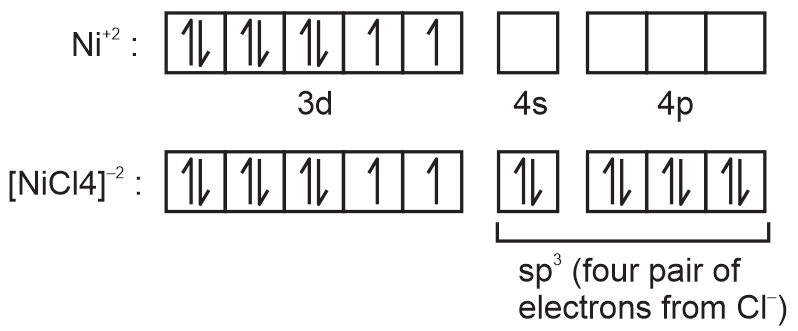
Geometry = Tetrahedral.