Engineering
Chemistry
Activity or Electrochemical Series
EMF Calculation and Galvanic Cell
Nernst Equation
Question
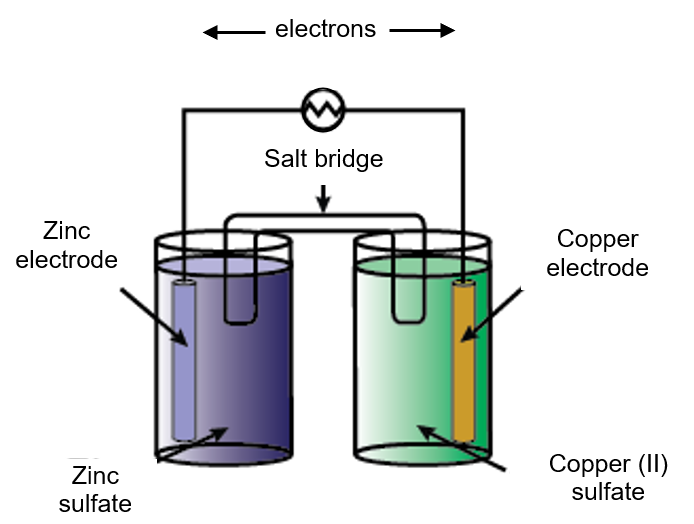
The given figure represents a Zn – Cu electrochemical cell:
Based on the figure, which of the following represents the balanced chemical reaction?
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
Zn(s) + Cu+2(aq.) → Zn+2(aq.) + Cu(s)
→ This is also called Galvanic cell reaction.
→ In this Cu+2 converts to Cu due to high standard reduction potentials than Zn.