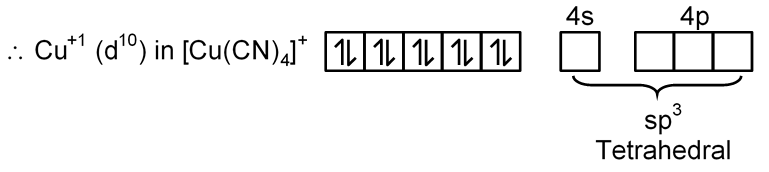The pair(s) of complexes wherein both exhibit tetrahedral geometry is(are)
(Note: py = pyridine Given: Atomic numbers of Fe, Co, Ni and Cu are 26, 27, 28 and 29, respectively)
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
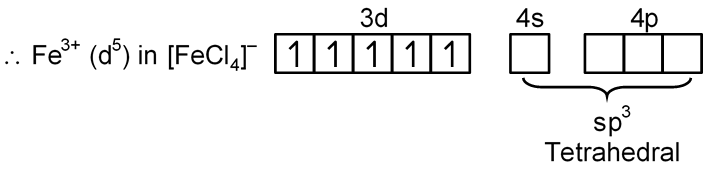
(A) [FeCl4]–
Fe → [Ar] 3d6 4s2
Fe+3 → [Ar] 3d5 4s0
Cl– is W.F.L. and does not pair up the unpaired electron of central metal atom.
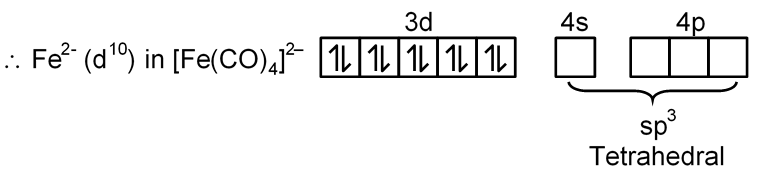
[Fe(CO)4]2–
Fe → [Ar] 3d6 4s2
Fe2– → [Ar] 3d8 4s2
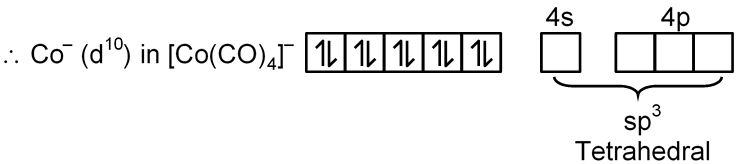
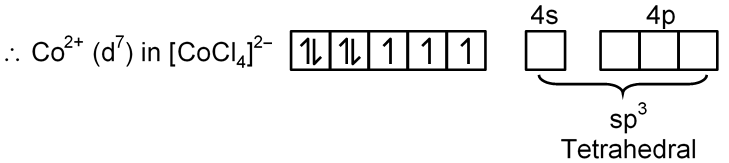
(B) [Co(CO)4]–
Co → [Ar] 3d7 4s2
Co–1 → [Ar] 3d8 4s2
[Co(CO)4]2–
Co → [Ar] 3d7 4s2
Co+2 → [Ar] 3d7 4s0
Cl– is W.F.L. and does not pair up the unpaired electron of central metal atom.
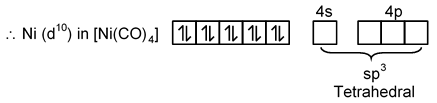
(C) [Ni(CO)4]
Ni → [Ar] 3d8 4s2
Ni0 → [Ar] 3d8 4s2
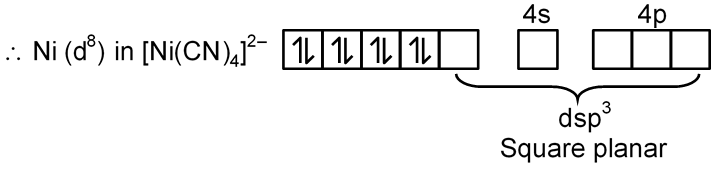
[Ni(CN)4]2–
Ni → [Ar] 3d8 4s2
Ni+2 → [Ar] 3d8 4s0
CN– is S.F.L. and pair up the unpaired electron of central metal atom.
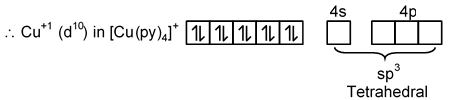
(D) [Cu(py)4]+
Cu → [Ar] 3d10 4s1
Cu+1 → [Ar] 3d10 4s0
[Cu(CN)4]3–
Cu → [Ar] 3d10 4s1
Cu+1 → [Ar] 3d10 4s0
CN– is S.F.L. and pair up the unpaired electron of central metal atom.