Engineering
Physics
Photoelectric Effect Einsteins Equation
Question
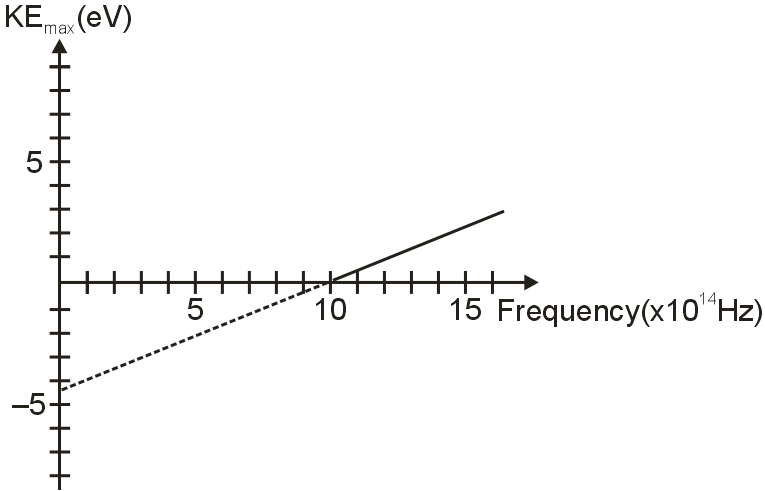
The pictured graph shows the relationship between the maximum kinetic energy of electrons flying off the surface of a metal versus the frequency of light striking the metal surface as part of the photoelectric effect.
According to this graph, what will happen when light of frequency 7 × 1014 Hz strikes the metal surface?
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
From figure, the threshold frequency of the metal vth = 10 × 1014 Hz
When light of frequency greater than the threshold frequency of the metal falls on the surface of metal, only then the photoelectrons are emitted. As the light used has frequency less that the threshold frequency of the metal, thus no electrons are emitted.
Hence option C is correct.