Question
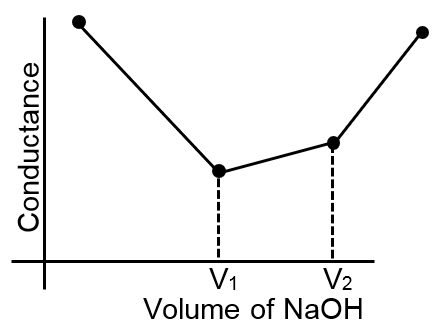
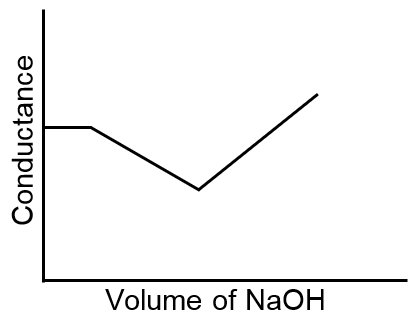
The principle of conductometric titrations is based on the fact that during the titration, one of the ions is replaced by the other and invariably these two ions differ in the ionic conductivity with the result that the conductivity of the solution varies during the course of the titration. Take, for example, the titration between a strong acid, say HCl, and a strong base, say NaOH. Before NaOH is added, the conductance of HCl solution has a high value due to the presence of highly mobile hydrogen ions. As NaOH is added, H+ ions are replaced by relatively slower moving Na+ ions. Consequently, the conductance of the solution decreases and this continues right upto the equivalence point where the solution contains only NaCl. Beyond the equivalence point, if more of NaOH is added, then the solution contains an excess of the fast moving OH– ions with the result that its conductance is increased and it continues to increase as more and more of NaOH is added. If we plot the conductance value versus the amount of NaOH added, we get a curve of the type shown in Fig 1.
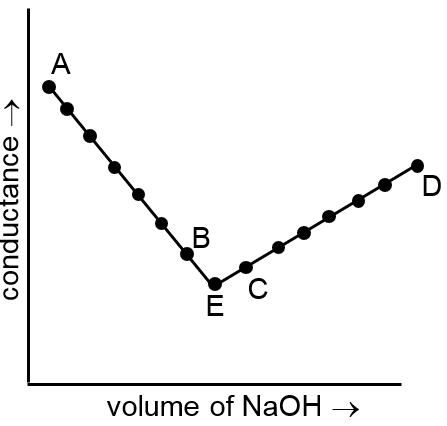
The descending portion AB represents the conductances before the euivalence point (solution contains a mixture of acid HCl and the salt NaCl) and the ascending portion CD represents the conductances after the equivalence point (solution contains the salt NaCl and the excess of NaOH). The point E which represents the minium conductance is due to the solution containing only NaCl with no free acid or alkali and thus represents the equivalence point. This point can, however, be obtained by the extrapolation of the lines AB and DC, and therefore, one is not very particular in locating this point experimentally as it is in the case of ordinary acid-base titrations involving the acid-base indicators.
Weak Acid versus Strong Base
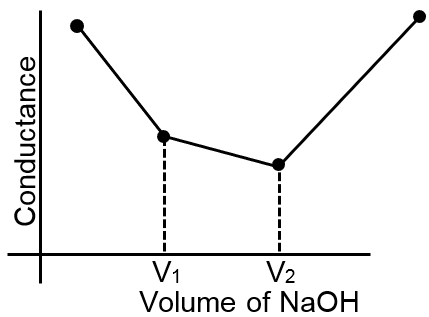
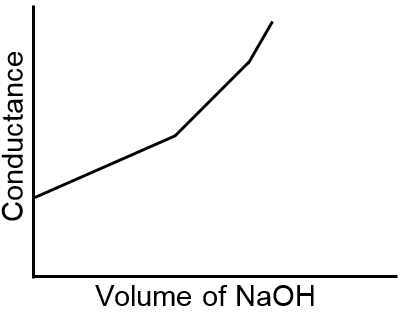
Let us take the specific example of acetic acid being titrated against NaOH. Before the addition of alkali, the solution shows poor conductance due to feeble ionization of acetic acid. Initially the addition of alkali casuse not only the replacement of H+ by Na+ but also suppresses the dissociation of acetic acid due to the common ion Ac– and thus the conductance of the solution decreases in the beginning. But very soon the conductance starts increasing as addition of NaOH neutralizes the undissociated HAc to Na+Ac– thus causing the replacement of non-conducting HAc with strong-conducting electrolyte Na+ Ac–. The increase in conductance continunes right up to the equivalence point. Beyond this point conductance increases more rapidly with the addition of NaOH due to the highly conducting OH– ions. The graph near the equivalence point is curved due to the hydrolysis of the salt NaAc. The actual equivalence point can, as usual, be obtained by the extrapolation method.
In all these graphs it has been assumed that the volume change due addition of solution from burrette is negligible, hence volume change of the solution in beaker the conductance of which is measured is almost constant throughout the measurement.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
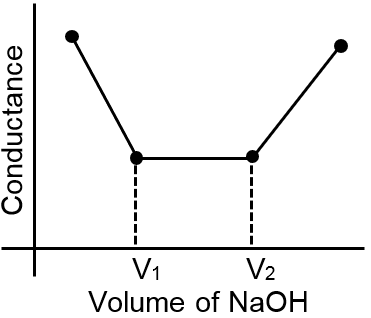
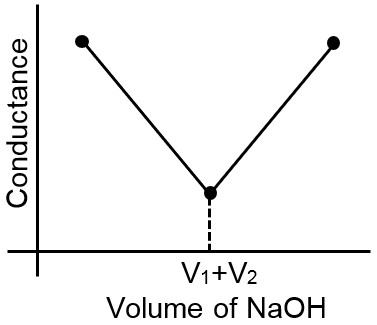
The most appropriate titration curve obtained when a mixture of a strong acid (say HCl) and a weak acid (say CH3COOH) is titrated with a strong base(say NaOH) will be
No solution
The nature of curve obtained for the titration between weak acid versus strong base as described in the above passage will be
No solution