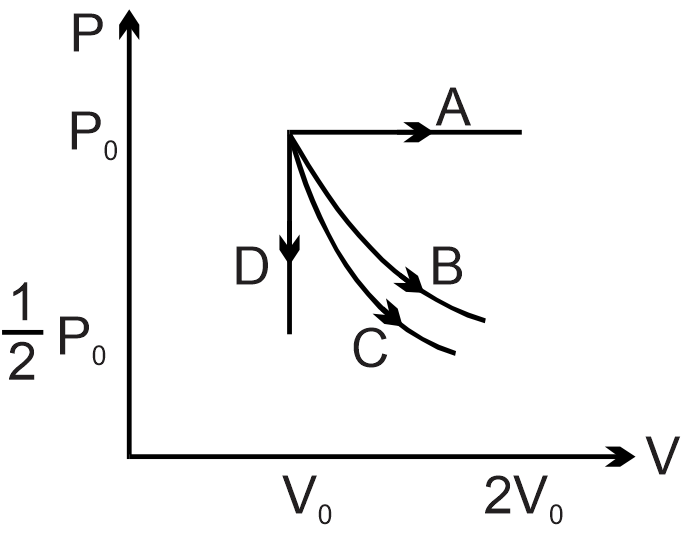
The PV diagram shows four different possible reversible processes performed on a monoatomic ideal gas. Process A is isobaric (constant pressure). Process B is isothermal (constant temperature). Process C is adiabatic. Process D is isochoric (constant volume). For which process(es) does the temperature of the gas decrease?
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
For an ideal gas, temperature change depends on work done and heat exchange. Process A (isobaric expansion): volume increases, so temperature increases. Process B (isothermal): temperature constant. Process C (adiabatic expansion): no heat exchange, work done by gas decreases internal energy, so temperature decreases. Process D (isochoric): volume constant, pressure decreases, so temperature decreases (from ideal gas law PV = nRT). Thus, temperature decreases only in processes C and D.
Final answer: Only Processes C and D