Engineering
Chemistry
Oxidation Number and Calculation
Compounds of Halogen Family
Empirical and Molecular Formula
Question
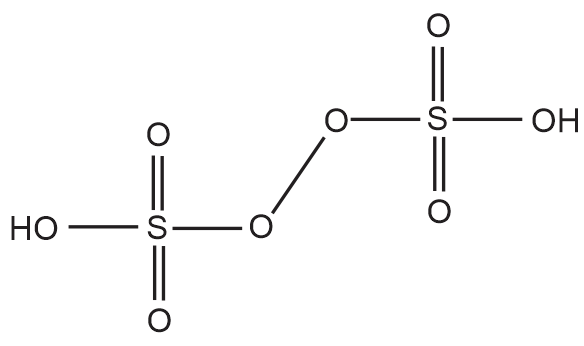
The ratio of oxygen atom having –2 and –1 oxidation numbers in S2O82– is _____.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
The ratio of oxygen atom having –2 and –1 oxidation numbers in S2O82– is three as only one peroxy linkage is present.
So, we can see from the structure below that the number of oxygen atoms having –2 oxidation state is 6 while those having –1 oxidation state is 2. Hence, the ratio is