Question
The relationship between pressure and volume of a gas was studied. A container was filled with helium gas at room temperature and sealed (see the diagram below). The pressure on the gas could be controlled by varying the force exerted on the plunger, and the volume could be determined by the height of the plunger.
(Note : Assume that the mass of the plunger is insignificant.)
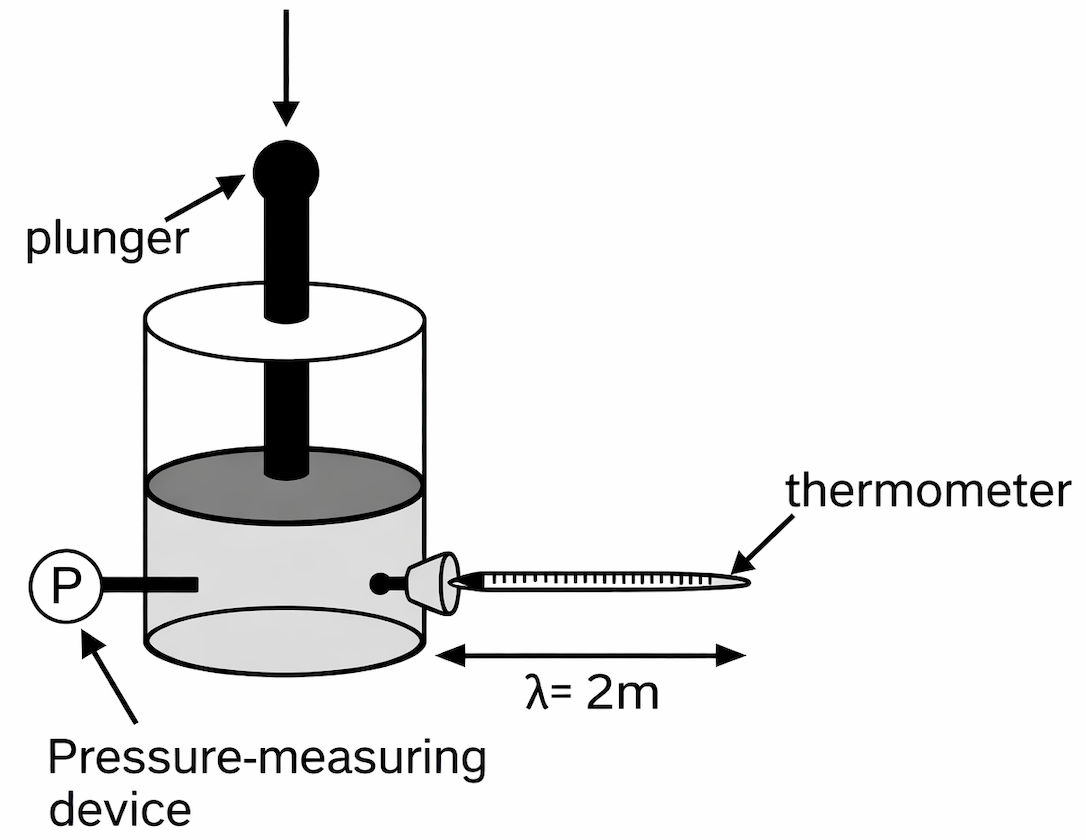
Experiment-1 :
The initial pressure in this experiment was 1 atmosphere (atm). At that pressure the gas occupied 1 litre (L). The pressure of the helium was increased very slowly, by decreasing the volume, so that the gas remained at room temperature. The heat generated was released into the surrounding atmosphere. The pressure and the volume of the gas helium were recorded in
Table-1
| Pressure (atm) | Volume (L) |
| 1.0 | 1.00 |
| 1.5 | 0.67 |
| 2.0 | 0.50 |
| 2.5 | 0.40 |
| 3.0 | 0.33 |
Experiment-2 :
Experiment 1 was repeated at room temperature with an initial pressure of 1 atm and an initial volume of 2 L. The results are shown in Table-2.
| Pressure (atm) | Volume (L) |
| 1.0 | 2.00 |
| 1.5 | 1.33 |
| 2.0 | 1.00 |
| 2.5 | 0.80 |
| 3.0 | 0.67 |
Experiment-3 :
In this experiment the entire gas container was insulated to prevent heat loss. The procedures from Experiment 1 were repeated. It was observed that the temperature of the gas rose as the volume decreased. Table-3 shows the volume measured at each pressure during the compression.
| Pressure (atm) | Volume (L) |
| 1.0 | 1.00 |
| 1.5 | 0.78 |
| 2.0 | 0.66 |
| 2.5 | 0.58 |
| 3.0 | 0.52 |
The insulation was then removed and the pressure maintained at 3 atm. As the gas cooled to room temperature, the volume of the gas slowly decreased from 0.52 to 0.33 L.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
How is the design of Experiment-1 different from that of Experiment-2?
No solution
Suppose that Experiment-3 was modified so that the insulation was removed after each pressure increase and the container was allowed to sit at that pressure until its temperature cooled to room temperature. How would the volume readings be influenced, if at all, by this modified procedure?
No solution