Engineering
Chemistry
Carbocation
Question
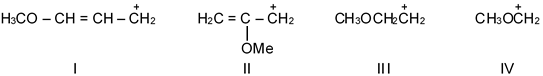
The relative stabilities of the following carbocations is :
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
Carbocation stability depends on hyperconjugation and resonance. More alkyl groups or resonance structures increase stability.
Carbocation I is tertiary and benzylic (most stable). Carbocation II is tertiary. Carbocation III is secondary but allylic (resonance stabilized). Carbocation IV is secondary (least stable).
Order: I (tertiary benzylic) > II (tertiary) > III (secondary allylic) > IV (secondary).
Final answer: I > II > III > IV