Engineering
Chemistry
Equilibrium Constant and Mass Action Law
Calculation of Various Parameters at Equilibrium
Arrhenius Equation
Question
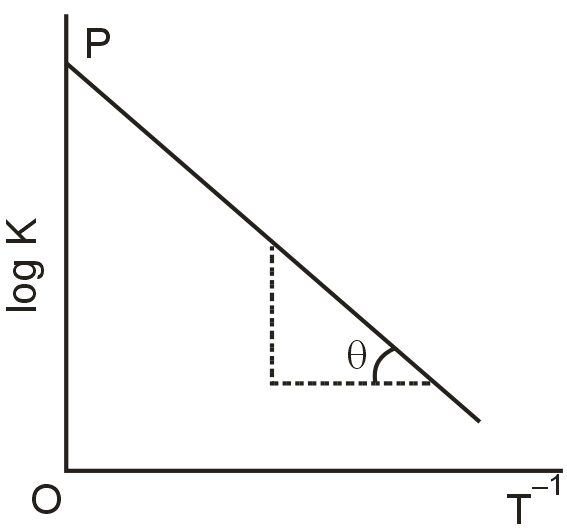
Variation of equilibrium constant K with temperature T is given by van't Hoff equation :
A graph between LogK and T–1 was a straight line as shown in the figure and having θ – tan–1 (– 0.5) and OP = 10. Calculate :
(a) ΔH° (Standard heat of reaction) when T = 298K
(b) A (pre - exponential factor)
(c) Equilibrium constant K at 298K
(d) K at 798K, if ΔH° is independent of temperature
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
(a)
It is an equation of a straight line of the y = c + mx
Slope
ΔH° = 9.574 mol–1
(b) Intercept D = log10 A = 10A = 1010
D = 1010
(c)
K = 9.96 × 109
(d)
log(K2) = 9.98 × 106