Engineering
Chemistry
Electronic Configuration
Crystal Field Theory and Valence Bond Theory
Colour and Magnetism
Question
What is the configuration of Cr+3 according to C.F.T. in aqueous medium?
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
In aquealy medium
ground state electronic configuration
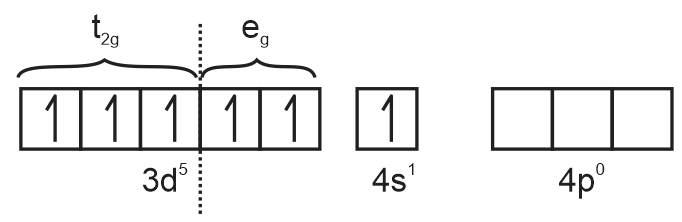
electronic configuration of Cr3+ =
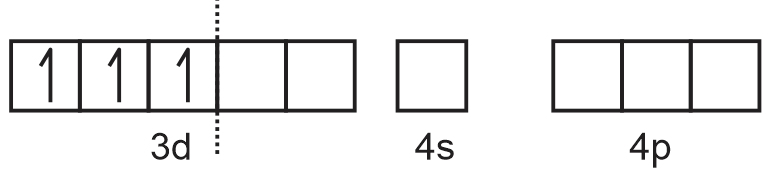
Formation of [Cr(H2O)6]3+ =
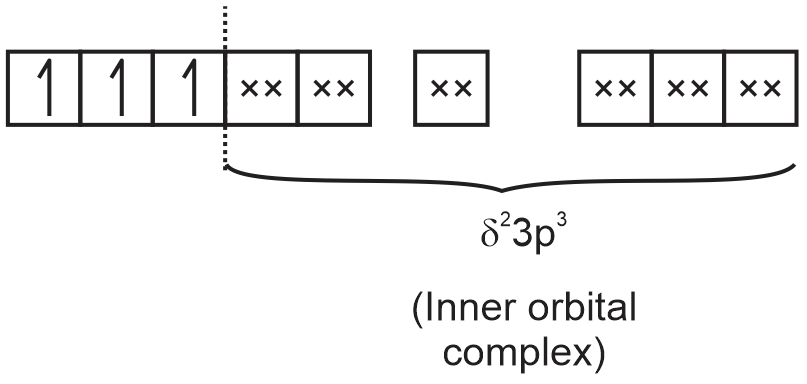
electronic configuration of Cr3+ according to C.F.T. = t32geg0
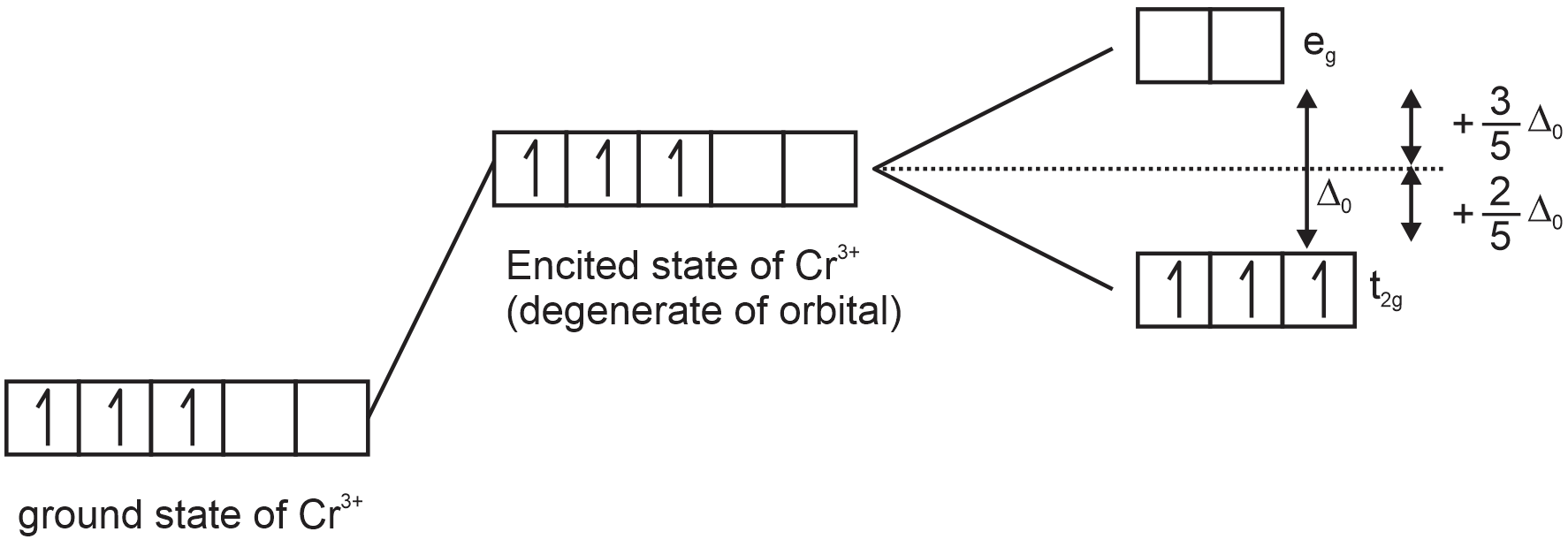
Here, no matter if the ligand is strong field or weak field the electronic configuration will going to stay the same,