Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
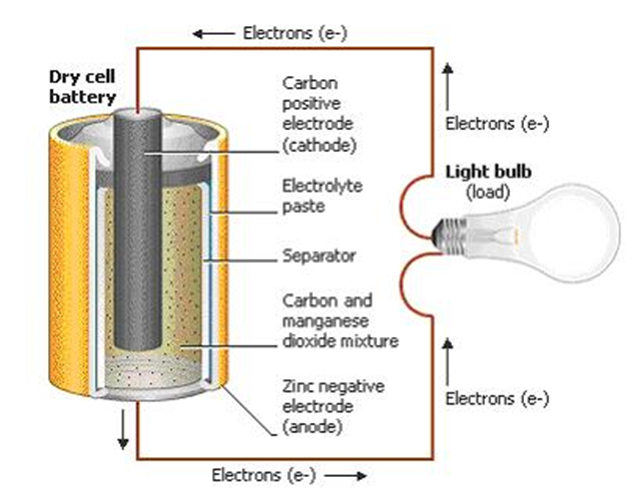
An electric battery is a device consisting of one or more electrochemical cells that convert stored chemical energy into electrical energy. Each cell contains a positive terminal, or cathode, and a negative terminal, or anode. Electrolytes allow ions to move between the electrodes and terminals, which allows current to flow out of the battery to perform work.
Cell convert chemical energy directly to electrical energy. Redox reactions power the battery. Cations are reduced (electrons are added) at the cathode during charging, while anions are oxidized (electrons are removed) at the anode during discharge. The electrodes do not touch each other, but are electrically connected by the electrolyte. A separator allows ions to flow between electrodes, but prevents mixing of the electrolytes.
The voltage developed across a cell's terminals depends on the energy release of the chemical reactions of its electrodes and electrolyte.
Hence, the chemical energy is converted into electrical energy.